Beyond genetics
Cancer and environmental factors

ABSTRACT
Cancer is one of the major health problems in the Western world given its high incidence and mortality rates. It is the second cause of death after cardiovascular disease, and the leading cause of mortality in people under seventy. This article takes a look at the external factors –chemical, physical and biological– that can trigger cancer.
Keywords: cancer, causes, environment, genetics.
Each year over ten million new cases of cancer are diagnosed worldwide, with a death toll of over six million a year. In Spain about 150,000 new cases are diagnosed yearly, of which over 70,000 people die.
The incidence of cancer continues to grow, and it is estimated that in 2020 over twenty million new cases will be diagnosed, with the most common being lung, colon, rectum, prostate and breast cancers.
In most cases, the exact cause of cancer is unknown. However, we do know that there are many factors able to cause a cell to abandon its natural cycle and start dividing uncontrollably. These factors can be internal (genetic) or external (environmental). However, they do not affect all people equally, as individual susceptibility plays an important role, so there are people exposed to these triggers who never develop a tumour.
«75-80% of tumours are caused by external, environmental factors and are therefore preventable»
Currently, cancer is considered to be a consequence of the accumulation of a series of genetic abnormalities. These can build up over decades, during which environmental carcinogens act by facilitating genetic changes.
It has been known for several decades now that 75-80% of tumours are caused by external –that is environmental– factors and are therefore preventable. Epidemiologists Doll and Peto (1981) made some calculations of the different factors involved in cancer deaths in the United States and the United Kingdom.
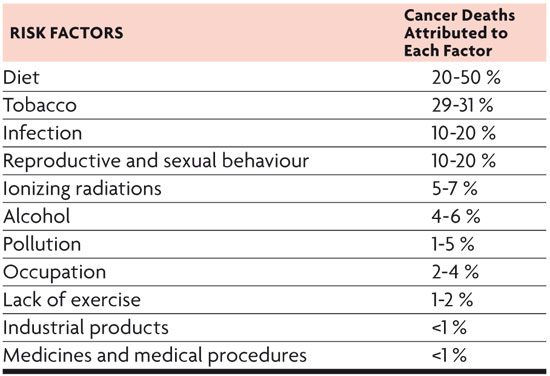
Quantitative estimation of risk factors favouring cancer development in the United States. / Table from Doll and Peto (1981).
CHEMICAL CARCINOGENS
The European Union defines chemical carcinogens as «substances and preparations which, if inhaled, swallowed or absorbed via the skin, may induce cancer or increase the frequency of this condition».
Carcinogenic substances are divided into three categories. The first includes substances with a cause/effect relationship between exposure to these and the development of cancer. The second category includes substances for which sufficient information is available to assume that human exposure to them can cause cancer. Finally, the third includes substances whose carcinogenic effects in humans are a cause of concern, but for which we do not have enough information to make a reasonable assessment. The main chemical carcinogens are: tobacco, alcohol, diet, asbestos and heavy metals.
Tobacco
Tobacco is the leading cause of mortality in general and is the chief cause of cancer in developed countries. Each year 20,000 new cases of lung cancer are diagnosed in Spain (Boyle, 1995). Tobacco is also responsible for cancers of the oropharyngeal cavity, oesophagus, bladder, urothelial tract and pancreas.
According to an official health survey conducted in Spain in 2006, 31.56% of men and 21.51% of women smoke. The 2009 European Health Survey gives similar figures for Spain. The 1990s witnessed an increase in women smokers; however, the trend does not seem to be ongoing.
«Tobacco is the leading cause of mortality in general and is the chief cause of cancer in developed countries. Quitting tobacco significantly reduces the risk of cancer five years later»
Tobacco smoke contains over 300 chemicals. According to the National Cancer Institute of the United States the number of chemicals is much higher, 300 are considered harmful to health, at least a quarter of them are carcinogens: nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone and polycyclic aromatic hydrocarbons such as benzopyrene induce mutations in the K-Ras protooncogene and p53 suppressor gene, respectively, altering DNA repair mechanisms.
The link between lung cancer and tobacco has been known since 1950 when Wynder and Graham (1950) published several studies showing a clear relationship between active smoking and lung cancer. People smoking tobacco with 22 mg or more of tar were found to be at a higher risk than if levels were below 21 mg.
The risk of developing cancer depends on the age at which one starts smoking and on nicotine content and the extent of inhalation. The 50 to 69 age range is when there is the greatest risk of developing cancer. Quitting tobacco significantly reduces the risk of cancer five years later. Fifteen years after quitting the habit this risk goes down by 80-90%, but it never becomes equivalent to that of non-smokers.
Passive smokers are also at an increased risk of developing cancer, and although it may seem small (1.5 times that of the general population) on the individual scale, at the population level it is a significant figure. In fact, environmental exposure to tobacco represents 25% of all lung cancers in nonsmokers (Hackshaw et al., 1997).

In Spain, about 30% of men and 20% of women smoke. Tobacco is the leading cause of cancer in developed countries and therefore one of the main factors to consider in cancer prevention campaigns. / Photo: SINC
Alcohol
A causal association has long been established between alcohol consumption and cancer of the oral cavity, pharynx, larynx, oesophagus, liver, colon, rectum and breast. Published data suggest that the effect of alcohol is modulated by polymorphisms in genes encoding enzymes involved in the metabolism of ethanol (alcohol dehydrogenase, aldehyde dehydrogenase, cytochrome p450) (Boffeta et al., 1990).
The exact mechanism by which alcohol exerts its carcinogenic effect is unknown but several have been proposed: genotoxic effect of acetaldehyde, elevated oestrogen concentration, production of reactive oxygen and nitrogen species, as well as changes in folate metabolism.
The risk of cancer increases with increasing levels of alcohol consumption. This conclusion was drawn from data taken from the study of 276,000 American drinkers (males) after a twelve-year follow up trial.
The risk of oral cavity and pharyngeal cancer increases in alcohol consumers, whether they smoke or not, but it seems that both factors together have a synergistic effect (Blot et al., 1988).
Alcohol also increases the risk of breast cancer. A study by the American Cancer Society, including over 500,000 women, followed up for twelve years, revealed an increased risk of breast cancer of about 20% in women who consumed one drink per day, and of 70% in those consuming five or more drinks per day. A meta-analysis of six case-control studies identified an increased risk of breast cancer only in women who drank more than three alcoholic beverages a day (Garfinkel et al., 1988).
Diet and exercise
Currently around 35% of cancers are attributed to diet-related factors. This association has been posed especially for tumours of the gastrointestinal tract (oesophagus, stomach, colon and rectum), and for tumours in areas sensitive to the action of sex hormones (breast, prostate, endometrium, ovary).
Gastric cancer appears to be associated with salted, smoked and nitrosamine-rich foods, which would seem to explain the high incidence of this tumour type in Japan and in Nordic countries. A study by Krukand Kladna published in 1999 confirmed that exercise early on protects against breast cancer. A meta-analysis of 43 studies reporting the relationship between weight and the time of breast cancer diagnosis showed a 33% increase in the risk of overall mortality and cancer-specific mortality in women who were obese at the time of diagnosis as compared to those who were not (Kruk and Kladna, 1999).

Six prospective studies, including more than 15,000 patients, have examined the relationship between physical activity and breast cancer prognosis. Five of them have shown a decrease in the risk of relapse and death in patients achieving moderate levels of physical activity after diagnosis. For example, research by the Nurses’ Health Study showed that patients who walked an average of three hours a week had a 50% decrease in the risk of breast cancer relapse, breast cancer-related death and death by any cause (Protani et al., 2010).
The incidence of colon cancer is linked to a diet high in animal fat and red meat and low in fibre. It would appear that fats favour increased bile-acid secretion and the growth of colonic bacteria able to metabolise these acids into carcinogens. Lower incidences of colon cancer have been observed in populations with higher fibre intake (Norat and Riboli., 2003).
In 1993 Dockery et al. published a prospective cohort study including 8,000 male participants, which showed a direct relationship between air pollution and lung cancer risk.
«A causal association has long been established between alcohol consumption and some types of cancer»
Occupational exposure to substances such as arsenic, asbestos or polycyclic aromatic hydrocarbons explains 18% of lung cancer cases in men and less than 1% in women. Other known risk factors include ionising radiation, chromium or nickel, and formaldehyde. Apparently they all act synergistically with tobacco. The effects of asbestos on health have been known for a long time; indeed, as far back as the first century Pliny the Elder described the disease in the lungs of slaves who wove asbestos cloth. A study published by Van Loon et al. in 1997 established a relative risk of 3.5 in people exposed to asbestos of developing lung cancer, adjusted for age and tobacco consumption. Cancer appears between 30 and 35 years after exposure to this cancerous agent.
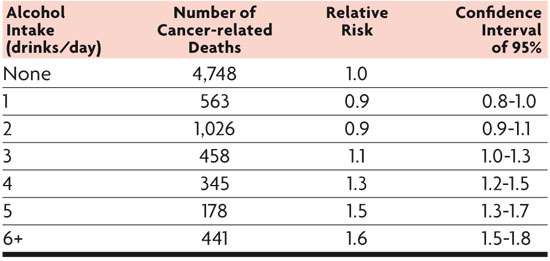
Relative risk of cancer mortality related to the number of alcoholic beverages consumed daily. / Data are from a cohort follow-up of 276,000 American men (Blot et al., 1992).
The mechanism by which asbestos causes cancer is not well defined, although the physical form of the mineral fibres is known to be important, the long and wide fibres being those with a greater capacity to cause cancer. Moreover, the iron these fibres contain may act as a catalyst in the production of hydroxyl-type free radicals, which cause DNA damage. Lung cancer is the most frequent tumour observed in workers exposed to asbestos, with tobacco having a clearly synergic effect, followed by mesothelioma (mesothelium cancer). The latter tumour is that most clearly associated with inhaling asbestos fibres, even though in 20% of diagnosed cases a connection with this fibre cannot be established.
Metals such as nickel, cobalt, cadmium or chromium are carcinogens that trigger the expression of hypoxia-inducible factor (HIF) and inhibit hypoxiarelated genes.
BIOLOGICAL CARCINOGENS
Viral infections are estimated to be responsible for one in seven human cancers. Around 80% are sequelae of hepatitis B (HBV) and hepatitis C (HCV) and human papillomavirus (HPV) (linked to hepatocellular carcinoma and cervical carcinoma, respectively). Epstein Barr virus has also been linked to the development of lymphoma and Helicobacter pylori with gastric cancer.
Prospective epidemiological studies have shown that people infected by the aforementioned virus have an increased risk of developing hepatocellular carcinoma (HCC), while chronic HBV infection is associated with an increase in liver cancer of almost 85% compared to uninfected controls. When there is HCV and HBV coinfection, then hepatocarcinoma incidence increases even more. Chronic inflammation, hepatocellular necrosis and cell regeneration taking place in HCV-relatedcirrhosis may be responsible for liver carcinogenesis. The risk of developing liver cancer depends on the intensity of infection; asymptomatic carriers have a lower risk of developing HCC than those with active hepatitis or cirrhosis.
«Ionizing radiation and ultraviolet radiation are also capable of causing cancer. Large-scale exposures such as those in nuclear disasters have made it possible to document this relationship»
In 2005 a meta-analysis was published of casecontrol studies on the additive effect of hepatitis B and C infection causing HCC in the Chinese population (Shi et al., 2005). The analysis included 32 studies with 3,201 cases and 4,005 controls from 1996 to 2004. The authors concluded that HBV and HCV infections are independent risk factors for HCC and that dual infection with both viruses is associated with an increased risk of developing this type of tumour.

This theory was confirmed by Donato et al., who conducted a review of epidemiological studies in southern Europe on liver cancer in 2006. They concluded that there is a synergistic effect between HBV and HCV infection, and added that alcohol can play a role as a cofactor of this disease.
The human papillomavirus (HPV) is responsible for 100% of cases of cervical cancer, and also causes 90% of anal cancers and 50% of cancers affecting the vulva and penis. This virus is transmitted sexually and types 16 and 18 are those responsible for the development of cervical cancer in 70-75% of cases.
PHYSICAL CARCINOGENESIS
Ionizing radiation and ultraviolet radiation are also capable of causing cancer. The carcinogenic mechanisms of ionizing radiation include damage to DNA. The incidence of cancer depends on the radiation dose. The most common tumours induced correspond to leukaemia, followed by some solid tumours such as lung or breast cancers. Ultraviolet radiation from the sun is closely associated with an increased risk of skin cancer, melanoma and non-melanoma.
The relationship between exposure to ionizing radiation and incidence of cancer has been known for many years. Large-scale exposures such as those in nuclear disasters like Hiroshima, Nagasaki and Chernobyl, have made it possible to document this relationship. The associated types of cancer are most often leukaemia and thyroid cancer; moreover, incidence varies with the dose, type and quality of the radiation received, as well as certain genetic factors (Pinar and Lara, 2004).

Ultraviolet radiation induces 90% of all skin cancers in humans, particularly nonmelanoma cancer. Therefore short but intense sun exposure, leading to skin burn, is related to an increase in the incidence of melanoma, whereas lengthy exposure, especially among farm workers and fishermen leads to an increase in non-melanoma skin cancer. A major factor in the risk of developing skin cancer is the amount of melanin, which protects us against UVA-B. Thus, people with white skin and red hair, who have less melanin, have a 50 times greater risk of developing skin cancer than those with dark skin.
Ultraviolet radiation damages DNA, producing pyrimidine dimers, which cannot be properly repaired in people with DNA-repair dysfunctions. Furthermore, immunity can be modified due to activation of suppressor lymphocytes. This immunodeficiency would lead to a high incidence of cancer in these patients.
To date, the relationship between exposure to electric and magnetic fields and cancer has not been demonstrated. Theoretically low-frequency fields, such as those surrounding power lines, cannot alter biological systems, since they lack ionizing power or the ability to sufficiently heat bodily tissues. The results of studies on the general population are contradictory, and analyses of studies on workers exposed to these fields have also proven negative. Similarly, there is no evidence, to date, of any relationship between cellphone use and an increased risk of developing brain tumours.
Bibliography
Blot, W. et al., 1988. «Smoking and Drinking in Relation to Oral and Pharyngeal Cancer». Cancer Research, 48: 3282-3287.
Blot, W. et al., 1992. «Alcohol and Cancer». Cancer Research, 52: 2119-2123.
Boffeta, P. et al., 1990. «Alcohol Drinking and Mortality among Men Enrolled in an American Cancer Society Prospective Study». Epidemiology, 1: 342-348. DOI: <10.1097/00001648-199009000-00003>.
Boyle, P., 1995. «Tabaco y cáncer. La perspectiva europea». Annals of Oncology, 7: 673-677.
Dockery, D. W. et al., 1993. «An Association between Air Polution and Mortality in Six US Cities». The New England Journal of Medicine, 329: 1753-1759. DOI: <10.1056/NEJM199312093292401>.
Doll, R. i R. Peto, 1981. «The Causes of Cancer. Quantitative Estimates of Avoidable Risks of Cancer in the United States Today». Journal National Cancer Institute, 66: 1291-1308.
Donato, F. et al., 2006. «Southern Europe as an Example of Interaction Between Various Environmental Factors: a Systematic Review of the Epidemiologic Evidence». Oncogene, 25(27): 3756-3770. DOI: <10.1038/sj.onc.1209557>.
Garfinkel, L.; Boffeta, P., & S. Stellman, 1988. «Alcohol and Breast Cancer: a Cohort Study». Preventive Medicine, 17: 686-693. DOI: <10.1016/0091-7435(88)90086-2>.
Hackshaw, A. K.; Law, M. R., & V. J. Wald, 1997. «The Accumulated Evidence of Lung Cancer and Environmental Tobacco Smoke». BMJ, 315: 980-988. DOI: <10.1136/bmj.315.7114.980>.
Harvey, E. et al., 1987. «Alcohol Consumption and Breast Cancer». Journal of the National Cancer Institute, 78: 657-661.
Kruk, A., & A. Kladna, 1999. «Sports Activity at Young Age Among Women After Mastectomy, compared to the control Group». Med Sport, 99: 29-33.
Norat, T., & E. Riboli, 2003. «Daily Products and Colorectal Cancer. A Review of Possible Mechanisms and Epidemiological Evidence». European Journal of Clinical Nutrition, 57(1): 1-17. DOI: <10.1038/sj.ejcn.1601522>.
Pinar Sedeño, B., & P. C. Lara Jiménez, 2004. «Carcinogénesis física». BioCancer Research Journal, 1.
Protani, M.; Coory, M., & J. H. Martin, 2010. «Effect of Obesity on Survival of Women with Breast Cancer: Systematic Review and Meta-analysis». Breast Cancer Research and Treatment, 123: 627-635. DOI: <10.1007/s10549-010-0990-0>.
Shi, J.; Zhu, L.; Liu, S., & W-f. Xie, 2005. «A Metaanalisis of Case-control Studies on the Combined Effect of Hepatitis B and C Virus Infections in Rausing Hepatocellular Carcinoma in China». British Journal of Cancer, 92(3): 607-612. DOI: <10.1038/sj.bjc.6602333>.
Van Loon, A. J. et al., 1997. «Occupational Exposure to Carcinogens and Risk of Lung Cancer: Results from The Netherlands Cohort Study». Occupation and Environmental Medicine, 54: 817-824. DOI: <10.1136/oem.54.11.817>.
Wynder, E. L., & E. A. Graham, 1950. «Tobacco Smoking as a Possible Etiologic Factor in Bronchiogenic Carcinoma. A Study of Six Hundred and Eighty-four Proved Cases.» The Journal of the American Medical Association, 143: 329-336. DOI: <10.1001/jama.1950.02910390001001>.





