«I am myself plus my circumstance, and if I do not save it, I cannot save myself», wrote Ortega y Gasset in Meditations on Quixote. Our most immediate biological «circumstance» is our microbiota, namely, the host of microbes living in and on our bodies. They cover every nook and cranny of our skin on the outside, while on the inside they coat our entire gastrointestinal tract, from our teeth and mouth through to the large intestine and anus. For nine months in our mother’s womb, we have no contact with microbes. However, from the moment of birth, we encounter not only a cold, dry and inhospitable environment, but also come into contact with microbes.
This contact, usually beneficial, will enable us to digest food and to develop an increasingly elaborate and effective immune system, in short, to live healthily. But we are also faced with germs that can cause diseases, and even lead to death. For many millennia of evolution, humans have developed a complex and intimate relationship with microbes. While we know they cause many infectious diseases, we now realise that the most representative and numerous interactions between microbes and other organisms are not pathogenic, but symbiotic.
«To the human body, the size of a mycoplasma is the same as the size of our body to the planet. The human body is the biosphere of our bacteria. A biosphere, which –like earth– harbours huge habitat diversity»
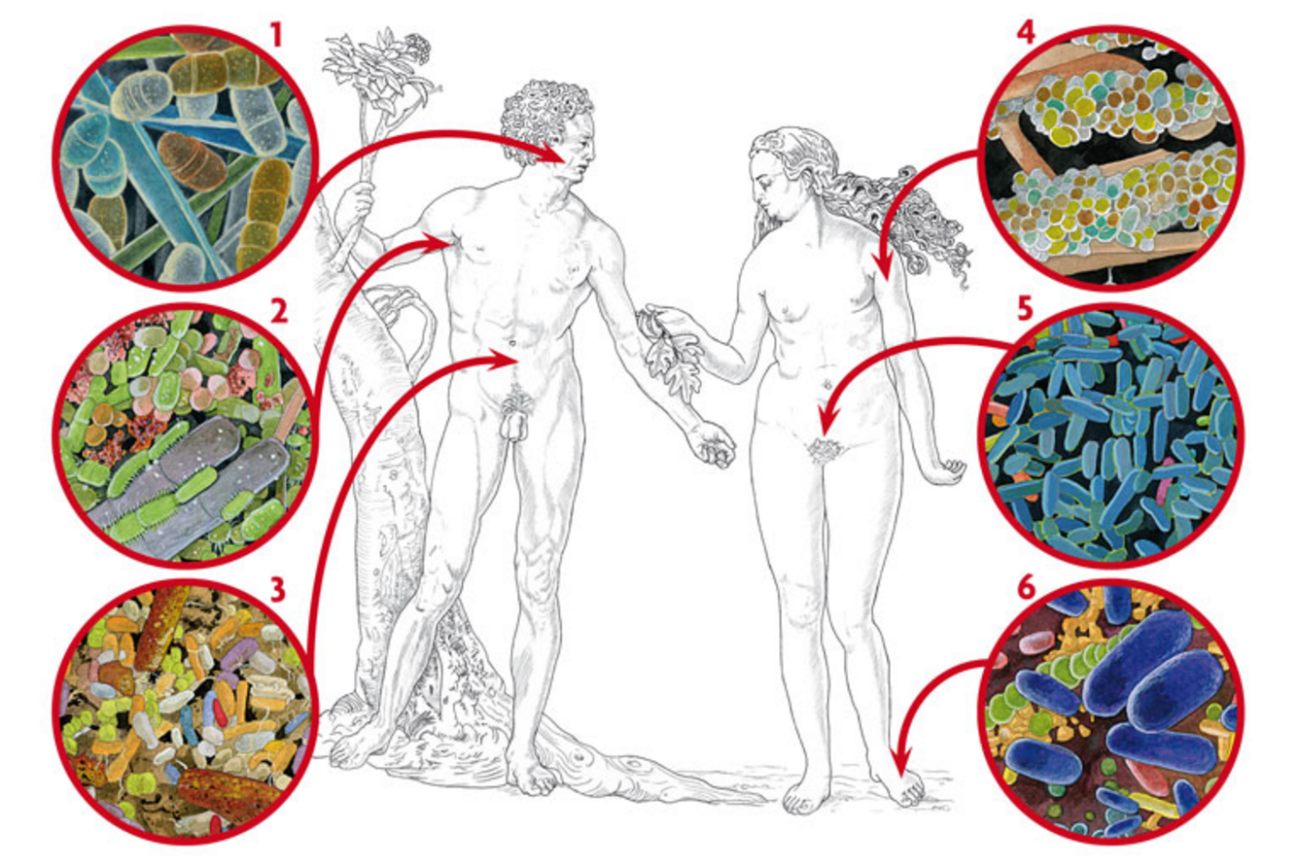
If we calculate the ratio between the height of an average person, for example 1.70 meters, and the diameter of the Earth, 12,756 km, it is about 7.5 °— 106. If we do the same by comparing a person’s height (1.70 m) with the size of the smallest free-living bacterium, Mycoplasma genitalium (which is a small sphere measuring about 0.225 μm in diameter), the ratio obtained is the same: 7.5 °— 106. Thus to the human body, the size of a mycoplasma is the same as the size of our body to the planet. The human body is the biosphere of our bacteria. A biosphere, which –like Earth– harbours huge habitat diversity. All bodily surfaces are colonised by microbiota: the forehead’s arid desert, the forearm’s cold forests, the rainforests of the armpit, the stomach’s acid lakes, the dense swamps of both intestines –large and small– full of food and lacking in air, the biofilms of fermenting lactobacilli in the vagina…
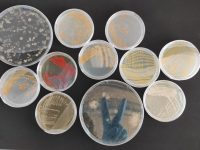
On its journey from birth to death, our body is colonised by many types of microbes: viruses, bacteria and fungi. An adult can have ten times more microbial cells than human ones: the human body has ten billion eukaryotic cells, «ours», and a hundred billion prokaryotic cells. In fact, it is estimated that all in all our body’s bacteria weigh 1.25 kg, representing 10% of our dry weight. As a whole, the genetic matter of the microbes inhabiting our body is called the microbiome. The number of genes belonging to the beneficial bacteria harboured by our bodies far exceeds the number of genes we inherit from our parents. Humans have about 23,000 genes. The intestinal tract’s microbiome alone amounts to 3.3 million genes. All these genes –belonging to «another» but ours too– enable us to perform basic processes such as digestion, synthesis of vitamins and amino acids, and maturation of the immune system.
Among humans, the microbiota composition is well preserved in terms of phyla (which are the major taxonomic divisions in bacteria), and is highly variable in terms of genera and species. Despite this variability, there is substantial conservation of microbiota functionality. The human microbiota belongs to less than ten of the fifty known bacterial phyla, and mainly to just six of them. A study of the microbiota inhabiting the palms of the hands of fifty-one healthy individuals found huge intra- and inter-personal diversity in the composition of bacterial communities. Most skin bacteria belong to four phyla: Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria. Cosmetics, soaps, hygiene products and moisturisers can contribute to the variation in microbiota of the skin.
An imbalance in microbiota may be responsible for chronic inflammatory diseases of the gastrointestinal tract, or associated with obesity. Obesity has become a widespread condition, of almost epidemic proportions, especially in developed countries. For this reason, efforts are being made to identify both environmental and host factors influencing the energy balance in these people. Comparative analyses of the human microbiome have revealed substantial changes in species composition of the microbiota and in the genes associated with a particular disease. A comparison of the intestinal microbiota of obese and lean mice, as well as of obese and lean human volunteers, has revealed that obesity is associated with changes in the relative abundance of two phyla of dominant bacteria: Bacteroidetes and Firmicutes. The proportion of Bacteroidetes decreases in obese subjects compared to lean ones; moreover, the proportion increases with weight loss caused by low-calorie diets. Metagenomic studies and biochemical analyses have found that the microbiota of obese individuals increases the capacity to exploit dietary energy. What is more, this feature is «transmissible»: when gnotobiotic or germ-free mice (experimentally bred without their natural microbes) are colonised with the microbiota of obese mice, the body fat of the former significantly increases. In contrast, when germ-free mice are inoculated with bacteria from a thin mouse they do not experiment an increase in body fat. These results clearly indicate that intestinal microbiota composition is an additional factor contributing to obesity.
At present, little is known about the mechanisms underlying the survival and long-term tolerance of indigenous microbial communities, or why these microbes do not produce a chronic or detrimental inflammatory response. Perhaps one of the greatest challenges facing human physiology, and microbiology, is to understand the temporal dynamics of biochemical communication between our body and its microbiota. And not only on the time scale of a human life, but also on an evolutionary time scale, in relation to the global changes in our diet and the environmental stressors of our species. Nowadays, it is essential to try to understand how changes in a person’s microbiota can be related to specific diseases, and to investigate the best way to therapeutically manipulate microbiota to obtain human health benefits.