Life in extreme conditions
The paradox of Antarctic marine biodiversity

The study of pristine places is very important for learning about the state of the oceans before the impact of human beings. Due to the extreme environmental conditions of the Antarctic continental shelf – its distance from other continents, depth, and the weight of the continental ice – it offers us a great opportunity to better understand how a pristine ecosystem would normally be. In addition to a high level of biodiversity, Antarctic benthic organisms present patterns of demographic and spatial distribution that are different from the communities of the continental shelves in other seas and oceans of the world. This makes Antarctic benthic communities look, more than one might think, like the communities with the highest known biodiversity in the world.
Keywords: marine biodiversity, conservation, Antarctica, benthos, gorgonia.
After more than a century of marine resource exploitation in all the oceans of planet Earth, treating them as humanity’s gutter or dustbin, there are very few places where the footprint of humans is not present. The effects or consequences of pollution, all sorts of waste, indiscriminate extraction of resources and, lately, climate change are clearly visible and are having different levels of impact in all the seas and oceans of the world. But there are still some places where, due to the distance from human activity, marine life remains almost intact or, at least, without the impact produced by humans. The islands in the middle of the Pacific Ocean or large and extensive abyssal zones are clear examples. But, carried by marine currents, waste or pollutants have reached even these latter, almost inaccessible, areas. The situation on the surface of the oceans is much more complicated, with the presence of the so-called islands of plastic.
The study of genuinely pristine locations is very important to improve our knowledge of their original status before none of them remain this way. One of the most important reasons to learn about how oceans were without the impact of humans is to know how the seabed is being degenerated or destructed. This is essential to develop more effective and appropriate restoration or recovery measures, so that we understand how the seabed should be and do not introduce artificial changes that could, quite possibly, generate even more alterations to the environment.
Why Antarctica?
Research on the most pristine seabeds on Earth led us to study life on the continental shelves of the high Antarctica – the most central areas of the great Antarctic seas – including the Weddell and Ross seas. These shelves are covered in sea ice most of the year (Lange, Ackley, Wadhams, Dieckmann, & Eicken, 1989). This layer, called the ice pack, is around two meters thick and has frozen and thawed every year for over 25 million years (Figure 1). For thousands of years, the persistence of the ice pack has preserved these areas from natural disturbances, as well as from the potential impacts of human activity. The ice pack extends beyond the outermost area of the shelves of the Antarctic continent, where glaciers regularly generate new icebergs, and it is these that produce the main natural impact on the seabed.
«The study of genuinely pristine locations is very important to improve our knowledge of their original status before none of them remain this way»
Icebergs detaching from the Antarctic continent glaciers are the main natural disturbance – almost the only one – that affects the Antarctic shelf seabed (Figure 2). The floating ice masses drag along the seabed for days or weeks, impacting long stretches and creating a complex habitat formed by a mosaic of disturbed and undisturbed areas. This partially or completely affected seabed is soon recolonised by nearby organisms, and this leads to the succession process. The early stages of this process are characterised by low abundance and diversity, and low substrate coating. The first colonisers usually grow quickly and have low structural complexity, while the final stages of the succession present high species richness and an exceptionally high biomass (Figure 3). These situations also occur in other types of ecological succession, for instance, the recovery of a forest area after a fire, on seabeds damaged after trawling, etc. Although it is estimated that only 5 % of the high Antarctic seabed has been affected by the action of icebergs in the last millennium (Gutt, 2000), the impact is very significant because the destroyed communities are millions of years old (Aronson & Blake, 2001). Any environmental phenomenon that can increase the usual iceberg release will have a crucial impact on the Antarctic seabed life. In this sense, the global increase in temperatures caused by global warming is the most significant phenomenon we must consider.

In the Antarctic survey ANTXXIX/9, carried out between 2013 and 2014 aboard the research ship Polarstern, previously inaccessible areas were reached in the south-east of the Weddell sea, specifically the Filchner-Ronne ice shelf. Taking advantage of this almost exceptional fact, studies were carried out on the 300-m deep seabed (Ambroso et al., 2017). The study used submarine robots that provided registered images along a 6 km-long path. The transects were recorded at locations that had never been explored and left no anthropogenic disturbances. The images showed a seabed that could be considered exceptional because of its biological richness. In a large part of the explored seabed surface, no area was free from sessile organisms (sponges, cnidarians, bryozoa, etc.) and the epibiont fauna was very rich, as were fish and crustaceans. Even though we knew from previous studies (Gutt & Starmans, 1998) that the benthic biodiversity – described as the richness of species living on the seabed – in the Antarctic continental shelves was one of the highest in all the oceans around the world, we were surprised to find so much diversity in a location that had been covered in ice for years (we found as much, or more, diversity than was then known for the entire Antarctic seabed).
«One of the most frequently asked questions is about the origin of this high diversity when the environmental conditions of this location are so extreme»
This study proved that, in addition to the high biodiversity, Antarctic benthic organisms present patterns of spatial and demographic distribution different from the communities of the continental shelves in other seas and oceans in the world, which have been affected – to a higher or a lesser degree – by anthropogenic disturbances. In Antarctica, the populations of sessile species such as gorgonians, sponges, or bryozoa, besides being quite high, presented individuals or colonies of different ages resulting from a long process of colonisation and growth (Figure 4). We can very easily guess that they are highly stable in their environment and have a low natural mortality. Therefore, the distribution patterns found in this study are a benchmark reference for the other communities. The more the distribution patterns of benthic organisms from other seas and oceans look like those found in the Antarctic seabed, the more we can consider them to be in a good state of conservation. Thus, studying the Antarctic seabed allows us to prove the great importance of learning about life in very pristine locations, so that we can explore that status of marine seabed shelves before the activity of humans altered them. In addition, it is very difficult to determine when or how a benthic community was impacted and whether what we are observing is a pristine environment or not. Having this Antarctic reference marks an important step towards a better understanding of medium-depth seabeds all over the world.
Why is the Antarctic seabed so diverse?
The Antarctic continent was partially isolated from the rest of the planet when the Antarctic circumpolar current was formed around 35 million years ago. The atmospheric circulation then changed dramatically, temperatures rapidly decreased, and the continent was covered in ice. With this ice cover, the flow of fine sediments carried by rivers stopped and, unlike the rest of continental shelves around the world, the Antarctic shelf was not covered in sediments. Thus, although the massive arrival of fine sediments eliminated a large part of the sessile organisms at the bottom of the shelf almost all over the world, Antarctic communities received very few sediments and what they did receive was larger and glacial. Thanks to this, benthic life has been able to survive for millions of years (Gili et al., 2006). Thus, Antarctic marine life has evolved unaffected by the last great oceanic extinction and has generated many species endemic to the Antarctic ocean. In fact, the level of endemism of Antarctic species is quite high: in some groups such as fish it is 90 % and in others it can reach over 50 % (Arntz, Brey, & Gallardo, 1994). The physical and environmental isolation, as well as its long isolated history, are the accepted explanations for this biogeographical phenomenon.

The high biodiversity of the Antarctic seabed is also explained by other environmental and biological factors (Figure 5). Among the former, the most important is environmental stability, with a constant water temperature between 1 and –1 °C throughout the year, for thousands of years. We must note the low presence of benthic species predators in the Antarctic ocean; this is because the low temperatures make it impossible for large predators such as decapod crustaceans or sharks, among others, to survive. The cold Antarctic waters are also loaded with nutrients, which allows for high rates of primary production, facilitating the development of many vertebrates in Antarctic waters.
«Marine ice is one of the most productive ecosystems in the world because a very unique ecosystem develops within»
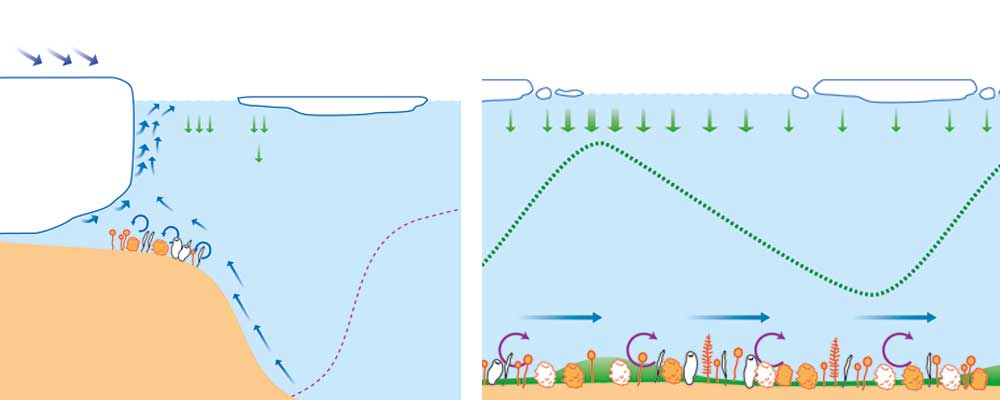
One of the most frequently asked questions is about the origin of this high diversity when the environmental conditions of this location are so extreme. Again, the low temperatures are one of the key factors. For millions of years, the Antarctic shelves have been covered by a layer of surface marine ice which stayed during the winter and melted in the summer; this ice is one of the most productive ecosystems in the world (Thomas & Dieckmann, 2002). The salt trapped inside it during the freezing process facilitates the formation of a system of millions of tiny canals in which a unique ecosystem develops: one dominated by microalgae (diatoms and other phytoplankton groups) surrounded by a rich community of bacteria and other microorganisms. Photosynthesis never stops; microalgae serve as food for the community of organisms around them, and these, through the products excreted by their own metabolism, generate nutrients that are essential to algae; thus, the ecosystem is constantly active despite the low temperatures. Because the algae in the ice have limited space, they cannot multiply, and so they accumulate large amounts of substances, especially lipids (many of them from the omega-3 group), and this also helps them to maintain the impermeability of their cell walls. For more than six months, the microalgae convert marine ice into a reservoir of one of the best foods that can be generated in the oceans. When spring comes to the southern hemisphere, the ice melts and the microalgae are freed from the ice. They usually then start to quickly form long chains and drop to the bottom of the sea relatively fast.
Thus, every spring, the Antarctic seabed is fertilised by tons of top-quality food, but because the temperature is very low, it does not decay (Figure 6). This food accumulated on the seabed forms what is known as «green carpets», which can feed the organisms at the bottom of the Antarctic shelves for months. But the chains of microalgae that drop down from the surface are not the only source of food for benthic organisms. When the ice melts, krill – at over a thousand million tonnes they are the most abundant invertebrate in the Antarctic ocean – consume large amounts of the phytoplankton freed from the marine ice. Krill feed so fast that they cannot digest half of what they eat, and so they excrete it as faeces (Arntz et al., 1994). These faeces quickly drop to the bottom and contain a lot of fresh microalgae that join those that had already been deposited on the seabed as more food for sessile organisms. Thus, the high biodiversity of Antarctic benthic communities can also be explained by the fact that, for millions of years, these communities have always had rich and abundant food that stays fresh for months because of the low temperatures. This constant food rain is a fundamental part of the relationship between plankton production and the benthic system, called benthopelagic coupling. The high availability and high quality of food comes with the low energy demands of many species, proved by their low oxygen consumption also resulting from the low temperatures. For instance, the oxygen consumption of Antarctic bivalve molluscs is one to two orders of magnitude lower than in temperate-sea species.

Antarctic benthic communities: Biodiversity, structure, and dynamics
Previous studies that compared Antarctic diversity with other regions of the planet showed evidence that some groups of organisms such as polychaetes, amphipods, or echinoderms, among others, present higher diversity and structural complexity in the Antarctic ocean than in other regions such as Hawaii or Great Britain (Clarke, Aronson, Crame, Gili, & Blake, 2004). In addition, the abundance of organisms is a remarkable in Antarctica: in biomass terms, we know that the Arctic and Antarctic oceans differ by one order of magnitude. Also of note is the longevity of the species that form the Antarctic benthic communities, although currently we only know the approximate age of some gorgonians, sponges, starfish, molluscs, and crustaceans. The slow growth rates, together with the long life-cycles of these Antarctic invertebrates, allow them to create highly structured mature communities. For instance, some gorgonians of the genus Thouarella have been estimated to be between 650 and 1,100 years old. Specimens recently collected in the high Antarctica are part of the current fauna and are the result of a long and complex evolutionary history (Martinez-Dios, Domínguez-Carrió, Zapata-Guardiola, & Gili, 2016).
From a spatial point of view, learning about the processes that affect the distribution, abundance, and interactions between species is one of the main lines of research in the ecology of Antarctic organisms. For example, the grouped distribution of most of the Antarctic gorgonian species is related to the presence of lecithotrophic larvae that facilitate their fast deposition after they are freed from the ice. In Thouarella variabilis, the simultaneous presence of two different-sized gametes has been observed both in male and female colonies, providing evidence for the presence of different generations at the same time. This enables a constant gametogenesis in which larvae are released throughout the year and, thanks to the continuous resuspension of organic matter deposited on the seabed in spring, the successful deposition of the larvae is possible. These species are also affected by epibenthic relationships (e.g., predation or fracture of their branches during silt deposition), so the most protected areas in the colonies are the ones that present the highest number of polyps in reproductive stages, while the polyps from the periphery are specialised in feeding.
«The high biodiversity of the Antarctic seabed is also explained by other factors such as environmental stability»
The epibenthic relationships established between epibionts and sessile fauna are key for the life-cycles of living organisms. This strategy allows competition for a space to be avoided and provides better access to the water column, which increases local diversity. It also makes benthic communities very complex and gives them a three-dimensional structure – one of the features of communities formed by benthic suspension feeders. Soft-seabed benthic communities from the Antarctic shelves are frequently characterised by their high diversity, biomass, and structural complexity. We can also find this in some tropical or temperate areas, where coral reefs or rocky seabed communities have a high density of organisms. This makes the Antarctic benthic communities look, more than one might think, like the communities with the highest known biodiversity in the world.
References
Ambroso, S., Salazar, J., Zapata-Guardiola, R., Federwisch, L., Richter, C., Gili, J-M., & Teixidó, N. (2017). Pristine populations of habitat-forming gorgonian species on the Antarctic continental shelf. Scientific Reports, 7(1), 12251. doi: 10.1038/s41598-017-12427-y
Arntz, W. E., Brey, T., & Gallardo, V. A. (1994). Antarctic zoobenthos. Oceanography and Marine Biology Annual Reviews, 32, 241–304.
Aronson, R. B., & Blake, D. B. (2001). Global climate change and the origin of modern benthic communities in Antarctica. American Zoology, 41(1), 27–39. doi: 10.1093/icb/41.1.27
Clarke, A., Aronson, R. B., Crame, J. A., Gili, J-M., & Blake, D. B. (2004). Evolution and diversity of benthic fauna of the Southern Ocean continental shelf. Antarctic Science, 16(4), 559-568. doi: 10.1017/S0954102004002329
Gili, J. M., Arntz, W. E., Palanques, A., Orejas, C., Clarke, A., Dayton, P., ... López-González, P. J. (2006). A unique archaic epibenthic community of sessile passive suspension feeders in the high Antarctic. Deep Sea Research II, 53(8-10), 1029–1052. doi: 10.1016/j.dsr2.2005.10.021
Gili, J. M., Coma, R., Orejas, C., López-González, P. J., & Zabala, M. (2001). Are Antarctic suspension feeding communities different from those elsewhere in the world? Polar Biology, 24(7), 473–485. doi: 10.1007/s003000100257
Gutt, J. (2000). Some «driving forces» structuring communities of the sublittoral Antarctic macrobenthos. Antarctic Science, 12(3), 297–313. doi: 10.1017/S0954102000000365
Gutt, J., & Starmans, A. (1998). Structure and biodiversity of megabenthos in the Weddell and Lazarev seas (Antarctica): Ecological role of physical parameters and biological interactions. Polar Biology, 20(4), 229–247. doi: 10.1007/s003000050300
Lange, M., Ackley, S., Wadhams, P., Dieckmann, G., & Eicken, H. (1989). Development of sea ice in the Weddell sea. Annals of Glaciology, 12, 92– 96. doi: 10.3189/S0260305500007023
Martinez-Dios, A., Dominguez-Carrió, C., Zapata-Guardiola, R., & Gili, J. M. (2016). New insights on Antarctic gorgonians’ age, growth and their potential as paleorecords. Deep Sea Research Part I: Oceanographic Research Papers, 112, 57–67. doi: 10.1016/j.dsr.2016.03.007
Thomas, D. N., & Dieckmann, G. S. (2002). Antarctic Sea Ice–A habitat for extremophiles. Science, 295, 641–644. doi: 10.1126/science.1063391





