Diet, drugs, and the brain
Are ultra-processed foods a gateway to addiction?

Foods that are rich in fats and sugars are pleasurable because they stimulate our reward circuits, the same circuits that are activated by drugs. In a context in which unhealthy diets and drug abuse are common from adolescence, it is important to investigate their consequences. This article reviews the relationship between especially tasty food, our brain’s reward system, and drug use. Studies with animal models have proven that an intermittent high-fat diet during adolescence increases the consumption of cocaine and ethanol. Moreover, recent research has shown the fundamental role of the diet in the development and treatment of addictions.
Keywords: high-fat diet, sugar, pleasure, addiction, drugs.
An innate preference that makes us fatter
Eating is not only essential for survival; it is also one of the main pleasures for humans. In the last few decades, the incidence of obesity has dramatically increased in developed countries. So much so that it is commonly referred to as the twenty-first century epidemic (James, 2008). To try to reverse this trend and reduce its impact on the economy and healthcare system, the number of basic and applied studies has increased in parallel. The main factors contributing to the increase of obesity cases in our society are a higher consumption of ultra-processed foods and a reduction in calorific needs, resulting from an increasingly sedentary lifestyle. For most of our evolutionary history, humans have not eaten abundantly, and have occasionally suffered from hunger. Our body developed in a context of nutritional scarcity and considerable energy expenditure, in which an innate preference for high-calorie foods (fat and sugar) improved the chance of survival during periods of famine. However, this evolutionary adaptation is not very advantageous in today’s society, where food is ever-present and access to it is guaranteed (Gold, 2011).
«Our body developed in a context of nutritional scarcity and considerable energy expenditure, in which an innate preference for high-calorie foods improved the chance of survival during periods of famine»
To answer why we eat – and most importantly, why we overeat – it is essential to distinguish between the homeostatic and hedonic signals regulating food intake. On the one hand, eating a meal rich in fat, sugars, and carbohydrates provides some of the nutrients necessary for the correct functioning of our body and is, at the same time, very pleasant. The homeostatic control of our food intake is governed by the energetic and metabolic balance and is activated by a deficit in caloric intake or energy reserves. The hedonic pathway, however, does not depend on nutritional needs, and prevails over the homeostatic pathway, leading us to consume even when sated. This pathway depends on the brain’s reward pathway and is responsible for our finding high-fat and high-sugar foods more palatable.
Our «pleasure eating» is mainly regulated by the mesolimbic pathway in the brain’s reward system, a neural circuit generating and providing pleasure in relation to some experiences. All basic life maintenance activities (feeding, sleeping, and reproducing) are pleasurable because they produce a release of dopamine in the nucleus accumbens, the main axis of the reward system. This is a highly successful evolutionary mechanism aimed at ensuring survival, because it will lead us to repeat the behaviours we naturally find pleasurable.
«We must consider the variable of nutritional habits when looking into the potential to develop an addiction and of its treatment»
Eating, of course, is one of these inherently rewarding activities. Today, thanks to advances in neuroimaging techniques, we can observe what happens in the brain in vivo when we eat foods with high fat or sugar content. Functional magnetic resonance studies show that, in healthy humans with a normal weight, consuming high-fat or high-sugar food causes a release of dopamine in the mesolimbic pathway. Furthermore, other food-related stimuli like photographs associated with food consumption, certain locations, emotional states, or people, can also activate brain areas related to the reward system (Volkow et al., 2013).
Tasty food, drugs, and the brain
Beyond individual preference, we are much more likely to binge on junk food than on vegetables and legumes. Potentially addictive substances, like cocaine and other drugs, are often the result of manipulations and refining processes that boost their addictive properties. This refining process is comparable to that of ultra-processed foods (Ifland et al., 2009). We can find many high-sugar (honey has up to 85 % sugar content) and high-fat foods in nature (such as hazelnuts, at 61 %), but the combination of high concentrations of both nutrients in the same product is rare. However, in the current context, the food industry has paid close attention to an evolutionary heritage that pushes us towards highly processed foods, so they can offer artificially tasty foods that are very pleasurable to our palate, but not at all beneficial to our health.
The consequences of continuously enjoying tasty food are not limited to being overweight or suffering from obesity and other associated conditions (diabetes, arteriosclerosis, hypertension, etc.). Stimulating our reward system repeatedly with waves of dopamine modifies the functioning of our brain in a similar way to the action of drugs. But, as with substance abuse, the mere increase in dopamine release cannot explain the difference between normal food intake and compulsive and excessive consumption.
Previous studies point to dopamine as a modulator of feeding behaviour and of the reinforcing effects of high-fat food. However, the opioid and the endocannabinoid systems play a crucial role in this interaction (Kelley & Berridge, 2002). While the opioid system is connected to hedonic food properties and modulates the release of dopamine when food is consumed, the endocannabinoid system is more closely connected to homeostatic control of the intake and a positive feedback to the consumption of high-fat food rather than carbohydrates or sugar. In fact, studies in animal models have found changes in the gene expression of opioid and cannabinoid receptors depending on the type of access to high-fat food (binging or continuously eating), as is the case with abuse drugs (Blanco-Gandía, Cantarcorps, et al., 2017; Blanco-Gandía, Ledesma et al., 2017; Mahler et al., 2007; Volkow et al., 2013).

The main factors contributing to the increase in obesity cases in our society is the higher consumption of ultra-processed foods and the reduction in caloric needs resulting from an increasingly sedentary lifestyle. / Freepik
Three distinct stages can be distinguished in the development of addiction: the escalation in consumption (and excessive consumption at specific times, or binge eating), dependence on the substance and, finally, craving and relapse in consumption. Similar to addictive processes, the moderate desire to consume ultra-processed food can become an excessive and compulsive pattern, accompanied by craving and even abstinence symptoms from certain components, such as sugar. Over time, drug abuse causes permanent brain changes, and the individual is still vulnerable to relapse even after a prolonged period of abstinence. In fact, this agrees with the commonly accepted definition of addiction as a chronic multifactorial disease characterised by a loss of control and relapse of substance abuse (Koob & Volkow, 2010).
Thus, as is the case with other substances, several phases have been described in the research of tasty food and binge eating, including the «like» phase and the «want» phase (Hoebel et al., 2009). «Liking» is related to the hedonic component (Berridge, 2009), while «wanting» is connected to the psychological process that provides a relevant stimulus and motivates us towards the consumption of something pleasant. For people who binge eat, they first consume because they like the substance; then, little by little, the craving for tasty foods starts in response to negative emotional states. Finally, this continued behaviour stimulates the reward system and turns the reward into a need rather than preference (want, rather than like), thereby gradually developing the addictive process (Robinson & Berridge, 2008). There is a concurrent learning process that generates stimuli associated with the reward and which anticipates or predicts the pleasurable response. As a consequence, the individual will feel an urgent need to consume again to obtain pleasure, thus compromising their decision-making ability (Berridge, 2009). In addiction models for abuse drugs, this hijacking of the reward system progressively prioritises consumption over any basic need.
It is a recognised fact in diagnostic manuals such as the DSM-5 (Diagnostic and Statistical Manual of Mental Disorders) and ICD-11 (International Classification of Diseases) that addictions are not limited to chemical substances. Any daily activity that produces pleasure can lead to addictive behaviours (for example, gambling). But, can we include the consumption of ultra-processed food in this definition of addiction? Scientific consensus has still not been reached on this matter, but some authors have suggested that one can get addicted to certain compounds in food (such as sugar, for instance), while others suggest that we should focus on the act of eating (Fletcher & Kenny, 2018).
«Potentially addictive substances, like cocaine and other drugs, are often the result of manipulations and refining processes that boost their addictive properties»
In today’s society, both drug use and junk food consumption are widespread, especially among the adolescent population. Adolescence is a critical development period that brings many changes at the physiological, cognitive, behavioural, and psychosocial levels. In the adolescent brain, the prefrontal cortex responsible for decision-making, planning, and inhibitory control is still immature, so impulsive and risky behaviour is more frequent at this stage. During this life stage, excessive consumption of junk food, combined with large amounts of alcohol, are common, especially during the weekend (Bisetto Pons et al., 2012).
In a context in which incorrect eating habits are frequent and drug use is widespread from an early age, the causal relationships between them, as well as their interactions and consequences, must be studied. We must consider the variable of nutritional habits when looking into the potential to develop an addiction and its adequate treatment.
Gateway theory
Both drug addiction and binge eating disorders have high comorbidity levels (Swanson et al., 2011) and are characterised by a loss of control in relation to consumption behaviour, dependence, and cravings. In fact, studies in humans indicate that we are much more likely to develop one of these disorders when another disorder has already been diagnosed (Flores-Fresco et al., 2018); in both cases there are alterations in appetite and satiety, as well as self-destructive and impulsive behaviours.
Because abuse drugs and the consumption of ultra-processed foods act on the same brain mechanisms, we wonder if excessive and chronic consumption of certain high-fat and high-sugar foods can stimulate the reward system, increasing vulnerability to drug use. Considering these relationships, the gateway theory has been proposed for food and substance abuse. This theory suggests that compulsive eating could facilitate the development of other maladaptive behaviours such as drug abuse (Blanco-Gandía, Cantarcorps et al., 2017; Degenhardt et al., 2009; Puhl et al., 2011).
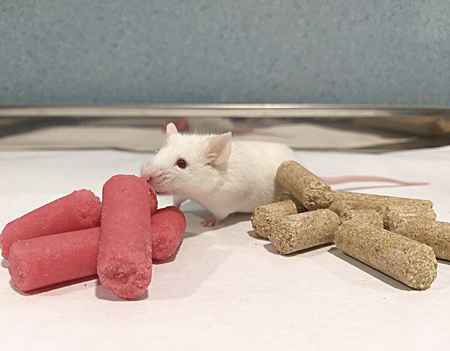
A special, high-fat (at least 45 %) and high-sugar feed is used to study the effect of the so-called cafeteria diet on rodents. Mice prefer to consume this food rather than what we would consider a balanced diet. / Macarena González
Among all the factors contributing to increasing vulnerability to drug use, the nutritional state is already recognised as a critical factor. Addiction studies carried out in animal models have made great advances in elucidating this interaction. They have examined the type of diet (what we eat) and the pattern of consumption (how we eat it), as well as how both of them modulate the development of drug addiction (Avena et al., 2004). In our laboratory, we observed that binging on high-fat food can stimulate the reward system, making mice more vulnerable to cocaine and alcohol consumption (Blanco-Gandía, Cantarcorps et al., 2017; Blanco-Gandía, Ledesma et al., 2017). Transferred to an experimental laboratory setting, binge eating consists of short periods of time (between one and two hours) that are repeated intermittently (three times a week, for instance) during which the animal has access to large amounts of tasty food, defined as a food item that is pleasurable to the specimen (in our studies, it is a high-fat – around 45 % – and high-sugar feed).
To check the effects that this food has on drug consumption, we compared the behaviour of mice that had been fed with different diets from an early age. We used two of the main models for evaluating the reinforcing effect of drugs: conditioned place preference and the self-administration paradigm.
For the conditioned place preference, we used a box with two different compartments with different colours (white/black) and textures (smooth/rough), separated by a neutral intermediate compartment. For four days, each mouse received a dose of alcohol or cocaine in one of these compartments to make them associate the rewarding effect of the drug with that particular setting. Therefore, this model evaluates the influence of environmental cues associated with the consumption of each particular drug. In these experiments, we observed that the animals that binged on tasty food during their adolescence spent more time in the compartment where they received the drug.
The self-administration paradigm is the reference procedure in rodents for evaluating the reward value of a drug. In this model, mice are trained to obtain the substance by performing an operant behaviour – in our case, a nose poke. Immediately after performing the task, the drug is infused to reward the behaviour. In this way, the animal gradually learns the association and repeats the behaviour to obtain the substance. In a series of experiments, we observed that animals intermittently exposed to high-fat food for two-hour periods consumed more alcohol than those that had a balanced diet.
The results suggested that this type of food could be enhancing their sensitivity to other stimuli, and we decided to study if the same was true for cocaine. As expected, the animals who had binged on fat since adolescence also self-administered more cocaine. Similarly, when we made the task required to obtain the drug more difficult (by increasing the number of required nose pokes), these animals showed more motivation to receive the drug (Blanco-Gandía et al., 2018). Not only did they consume more, but they also worked harder to get it.
«All basic life maintenance activities (feeding, sleeping, and reproducing) are pleasurable because they cause the release of dopamine»
But was the critical factor that increased the consumption of ethanol and cocaine the food itself, or the way in which it was consumed? To answer this question, we studied another group of mice with continued access to the high-fat diet. After two months, these mice were significantly heavier and perfectly represented the animal obesity and metabolic syndrome model. However, they did not show increased sensitivity to drugs (Blanco-Gandía, Aracil-Fernández et al., 2017). Meanwhile, other studies have shown that continued access to fat and sugar can be an alternative reward and decrease the reinforcing effects of drugs (Blanco-Gandía, Aracil-Fernández et al., 2017; Wellman et al., 2007).
Therefore, these studies suggest that the way tasty food is consumed is more important than its composition. When repeatedly exposed to binge eating, animals escalated the consumption of this type of food, eating more and more despite being sated, thus, disregarding their homeostatic satiety signals. This produces neuroadaptations in the reward system that have consequences not only on eating behaviour, but also on the modulation of their response to drugs and their vulnerability to addiction.
Final remarks
We must be aware that the type of food and the way we eat it are determining factors not only of our physical health, but also of our brain functioning and thus, our mental health. Children and adolescents are the main target of the processed food industry, which has been spending money for years to make their products even more appealing (for example, with gifts, flashy colours, or cartoon characters in their commercials). This makes it almost impossible to avoid being exposed to ultra-processed food at critical neurodevelopmental stages.

Children and adolescents are the main target of the processed food industry, which has been spending money for years to make their products even more appealing (for example, with gifts, flashy colours, or cartoon characters in their commercials). / Lucía Sapiña
Our studies prove that diet plays a fundamental role in both the acquisition and treatment of addiction. Research results to date highlight the fact that exposure to high-fat and high-sugar food in early years leaves a mark on the brain reward system. If, moreover, these functional and structural changes occur during critical developmental periods, the impact on the reward system will be permanent and these adolescents could turn into adults whose brain reward system is unbalanced and who are more vulnerable to the reinforcing effect of drugs.
It is important to note that the studies mentioned in these texts were performed in laboratory animal models and so their extrapolation to humans requires caution. Even though previous neuroimaging studies in humans indicate that binge eating activates the same neural networks underlying addiction – albeit to a lesser extent than drugs (Small et al., 2003; Volkow et al., 2008) – further clinical research is required to support this data.
References
Avena, N. M., Carrillo, C. A., Needham, L., Leibowitz, S. F., & Hoebel, B. G. (2004). Sugar-dependent rats show enhanced intake of unsweetened ethanol. Alcohol, 34(2), 203–209. https://doi.org/10.1016/j.alcohol.2004.09.006
Berridge, K. C. (2009). «Liking» and «wanting» food rewards: Brain substrates and roles in eating disorders. Physiology & Behavior, 97(5), 537–550. https://doi.org/10.1016/j.physbeh.2009.02.044
Blanco-Gandía, M. C., Aracil-Fernández, A., Montagud-Romero, S., Aguilar, M. A., Manzanares, J., Miñarro, J., & Rodríguez-Arias, M. (2017). Changes in gene expression and sensitivity of cocaine reward produced by a continuous fat diet. Psychopharmacology, 234(15), 2337–2352. https://doi.org/10.1007/s00213-017-4630-9
Blanco-Gandía, M. C., Cantacorps, L., Aracil-Fernández, A., Montagud-Romero, S., Aguilar, M. A., Manzanares, J., Miñarro, J., & Rodríguez-Arias, M. (2017). Effects of bingeing on fat during adolescence on the reinforcing effects of cocaine in adult male mice. Neuropharmacology, 113, 31–44. https://doi.org/10.1016/j.neuropharm.2016.09.020
Blanco-Gandía, M. C., Ledesma, J. C., Aracil-Fernández, A., Navarrete, F., Montagud-Romero, S., Aguilar, M. A., Manzanares, J., Miñarro, J., & Rodríguez-Arias, M. (2017). The rewarding effects of ethanol are modulated by binge eating of a high-fat diet during adolescence. Neuropharmacology, 121, 219–230. https://doi.org/10.1016/j.neuropharm.2017.04.040
Blanco-Gandía, M. C., Miñarro, J., Aguilar, M. A., & Rodríguez-Arias, M. (2018). Increased ethanol consumption after interruption of fat bingeing. PLOS One, 13(3), e0194431. https://doi.org/10.1371/journal.pone.0194431
Bisetto Pons, D., Botella Guijarro, A., & Sancho Muñoz, A. (2012). Eating disorders and drug use in adolescents. Adicciones, 24, 9–16.
Degenhardt, L., Chiu, W. T., Conway, K., Dierker, L., Glantz, M., Kalaydjian, A., & Kessler, R. C. (2009). Does the «gateway» matter? Associations between the order of drug use initiation and the development of drug dependence in the National Comorbidity Study Replication. Psychological Medicine, 39(01), 157–167. https://doi.org/10.1017/S0033291708003425
Fletcher, P. C., & Kenny, P. J. (2018). Food addiction: A valid concept? Neuropsychopharmacology, 43(13), 2506–2513. https://doi.org/10.1038/s41386-018-0203-9
Flores-Fresco, M. J., Blanco-Gandía, M. D. C., & Rodríguez-Arias, M. (2018). Alterations in eating behavior in patients with substance abuse disorders. Clínica y Salud, 29(3), 125–132. https://doi.org/10.5093/clysa2018a18
Gold, M. S. (2011). From bedside to bench and back again: A 30-year saga. Physiology & Behavior, 104(1), 157–161. https://doi.org/10.1016/j.physbeh.2011.04.027
Hoebel, B. G., Avena, N. M., Bocarsly, M. E., & Rada, P. (2009). A behavioral and circuit model based on sugar addiction in rats. Journal of Addiction Medicine, 3(1), 33–41. https://doi.org/10.1097/ADM.0b013e31819aa621
Ifland, J. R., Preuss, H. G., Marcus, M. T., Rourke, K. M., Taylor, W. C., Burau, K., Jacobs, W. S., Kadish, W., & Manso, G. (2009). Refined food addiction: A classic substance use disorder. Medical Hypotheses, 72(5), 518–526. https://doi.org/10.1016/j.mehy.2008.11.035
James, W. P. T. (2008). WHO recognition of the global obesity epidemic. International Journal of Obesity, 32(7), S120–S126. https://doi.org/10.1038/ijo.2008.247
Kelley, A. E., & Berridge, K. C. (2002). The neuroscience of natural rewards: Relevance to addictive drugs. Journal of Neuroscience, 22(9), 3306–3311. https://doi.org/10.1523/JNEUROSCI.22-09-03306.2002
Koob, G. F., & Volkow, N. D. (2010). Neurocircuitry of addiction. Neuropsychopharmacology, 35(1), 217–238. https://doi.org/10.1038/npp.2009.110
Mahler, S. V., Smith, K. S., & Berridge, K. C. (2007). Endocannabinoid hedonic hotspot for sensory pleasure: Anandamide in nucleus accumbens shell enhances «liking» of a sweet reward. Neuropsychopharmacology, 32(11), 2267–2278. https://doi.org/10.1038/sj.npp.1301376
Puhl, M. D., Cason, A. M., Wojnicki, F. H., Corwin, R. L., & Grigson, P. S. (2011). A history of bingeing on fat enhances cocaine seeking and taking. Behavioral neuroscience, 125(6), 930. https://doi.org/10.1037/a0025759
Robinson, T. E., & Berridge, K. C. (2008). The incentive sensitization theory of addiction: Some current issues. Philosophical Transactions of the Royal Society of London B: Biological Sciences, 363(1507), 3137–3146. https://doi.org/10.1098/rstb.2008.0093
Small, D. M., Jones-Gotman, M., & Dagher, A. (2003). Feeding-induced dopamine release in dorsal striatum correlates with meal pleasantness ratings in healthy human volunteers. Neuroimage, 19(4), 1709–1715. https://doi.org/10.1016/s1053-8119(03)00253-2
Swanson, S. A., Crow, S. J., Le Grange, D., Swendsen, J., & Merikangas, K. R. (2011). Prevalence and correlates of eating disorders in adolescents: Results from the national comorbidity survey replication adolescent supplement. Archives of General Psychiatry, 68(7), 714–723. https://doi.org/10.1001/archgenpsychiatry.2011.22
Volkow, N. D., Wang, G. J., Fowler, J. S., & Telang, F. (2008). Overlapping neuronal circuits in addiction and obesity: evidence of systems pathology. Philosophical Transactions of the Royal Society B: Biological Sciences, 363(1507), 3191–3200. https://doi.org/10.1098/rstb.2008.0107
Volkow, N. D., Wang, G. J., Tomasi, D., & Baler, R. D. (2013). Obesity and addiction: Neurobiological overlaps. Obesity Reviews, 14(1), 2–18. https://doi.org/10.1111/j.1467-789X.2012.01031.x
Wellman, P. J., Nation, J. R., Davis, K. W. (2007). Impairment of acquisition of cocaine self-administration in rats maintained on a high-fat diet. Pharmacology Biochemistry and Behavior, 88(1), 89–93. https://doi.org/10.1016/j.pbb.2007.07.008





