Numerous drugs with analgesic effect can be used to treat pain. Opioids are effective for moderate to severe pain, while nonsteroidal anti-inflammatory drugs and especially paracetamol (acetaminophen) are useful to treat mild or even moderate pain. Analgesics should be used according to guidelines, and users should be aware of their adverse effects, avoiding them whenever possible, and steering clear of overuse.
Drug therapy is a mainstay of the therapeutic approach to pain and includes not only the use of analgesics but also drugs to treat the sufferer’s affective factors. Drugs used to treat pain can be classified as: analgesic opioids, NonSteroidal Anti-Inflammatory Drugs (NSAIDs), simple analgesics and adjuvant drugs including local anaesthetics, antidepressants, anxiolytics, anticonvulsants, neuroleptics, corticosteroids, sympatholytic agents and an assorted group of substances without direct effect on pain itself but when used in combination with analgesics may improve the patient’s symptoms.
«In Spain, opioids are used less than in other nearby countries due to ignorance and fear of the potential risks involved»
Drugs used for treating pain
Opioid analgesics
The knowledge that opium can relieve pain and induce sleep is centuries old and has been put into practice by all civilizations. The main active component of opium is morphine, and since the seventies we have known that this –and other alkaloids extracted from opium– exert their action by stimulating specific receptors called opioid receptors. We use the term opiate to refer to analgesics made directly from opium, while opioid refers to a fully- or semi-synthetic alkaloid with the same mechanism of action.
These drugs are useful to treat any type of pain, and their use to combat acute pain and to treat cancer patients is well established; however, their prescription for non-neoplastic chronic pain is controversial, even though they are safe and easy to use. Years ago the use of opioids to treat non-cancer-related chronic pain was reserved for patients only when other pain relief methods failed; however, nowadays its use is accepted for any persistent pain that causes distress, disability or having a negative impact on the quality of life. Its main advantage is that it lacks an «analgesic ceiling», as increasing the dose increases the effect indefinitely. However, high doses cause adverse effects, such as respiratory depression, which in practice prevents the dose from being increased to the extent one may wish. Furthermore, they can lead to dependence.
In Spain, these are used less than in other nearby countries, which can be attributed to ignorance of their pharmacological characteristics and fear of the potential risks involved. Constipation is the most common adverse secondary effect and should be taken into account and prevented systematically, as many patients likely to use such medicines are elderly patients often prone to this clinical condition.
For correct prescription, a good recommendation is to follow the guidelines set for the clinical use of opioids in chronic non-malignant pain by the American Society of Interventional Pain Physicians.
NonSteroidal Anti-Inflammatory Drugs
For years NSAIDs have been the painkillers par excellence (taken whenever the patient feels pain) and are widely self-medicated. The mechanism underlying the anti-inflammatory action is to inhibit the synthesis of prostaglandins, endogenous pain and inflammation mediators. This mechanism is responsible for the effects common to all drugs within this group, both therapeutic (analgesic, antipyretic and anti-inflammatory) and adverse (gastrointestinal, renal…).
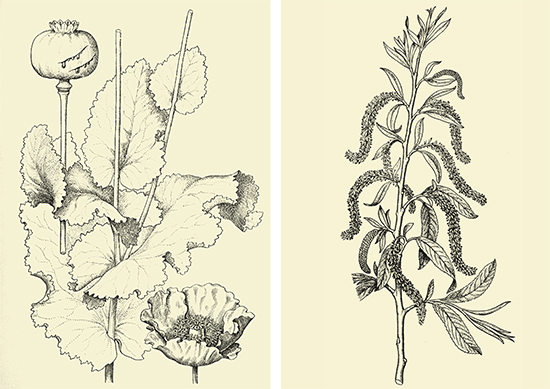
Opium’s potential to relieve pain has been known for centuries. On the left, The picture shows the opium poppy (Papaver somniferum) from Dioscórides renovado, Pius Font i Quer. One can see a flower and a capsule with slits made to extract the opium.
On the right, one of the most widely used drugs is aspirin, which is derived from the bark of white willow (Salix alba). Above picture taken from Dioscórides renovado, Pius Font i Quer. / © Biblioteca del Jardí Botànic de la Universitat de València
NSAIDs have moderate analgesic efficacy and are able to relieve a wide range of discomforts: headache, arthralgia, myalgia, tendinitis, dysmenorrhea, postoperative and posttraumatic pain, certain types of colic, early stages of cancer, and so on. The response is dose dependent, but they have an analgesic ceiling after which increasing the dose does not provide benefits but does imply greater risks of adverse effects (most commonly affecting the digestive system, kidney, or are of the allergic kind).
Related gastrointestinal complications are due not only to local irritation but also a systemic effect, which explains why they may cause injury even when administered by routes other than oral.
There are risk factors that make patients prone to complications,1 but pharmacological prophylaxis is possible in high-risk patients through the use of prostaglandin analogues, such as misoprostol, histamine H2-receptor blockers, like ranitidine, or proton pump inhibitors (PPI), such as omeprazole.
Simple analgesics
Within this group are substances with analgesic properties similar to those described as NSAIDs, but without an anti-inflammatory effect. These include paracetamol, metamizole or aspirin in low doses.
Paracetamol has a significant analgesic effect but lacks both anti-inflammatory and gastric-related effects, thus it is thought to have different mechanisms of action to NSAIDs. It is considered the antipyretic and analgesic drug of choice, preferable to NSAIDs, especially preferred over aspirin as it causes far fewer adverse reactions. However, there are risks of adverse effects as it can cause allergic reactions –usually in the form of urticaria or less frequently blood disorders like leukopenia or thrombocytopenia–. The most important drawback is the risk of intoxication, which can lead to severe liver necrosis and appears when adults exceed 7.5 grams and children exceed 150 mg per kilo per day.
Metamizole also lacks an anti-inflammatory effect and causes less gastric irritation than NSAIDs. Its analgesic ceiling is slightly higher and is similar to that achieved with low doses of opioids. It is also a smooth-muscle relaxant, making it useful to treat cramps; however, it can cause adverse kidney reactions and hypersensitivity. The most serious side effects are hematologic conditions (agranulocytosis and aplastic anemia), although the risk is very low.
Aspirin or acetylsalicylic acid has been regarded as one of the top NSAIDs. However, at the dosage normally taken, it has virtually no anti-inflammatory effect, which requires doses of between 3 and 6 grams per day. The antipyretic and analgesic effects of aspirin are obtained with doses of between 325 and 650 mg every 6 hours in adults. Furthermore, it has an antiplatelet effect at low doses (75-325 mg/day) and is thus used to prevent myocardial infarction in patients at risk. The most common adverse reactions fall into the gastrointestinal category, followed by kidney conditions and hypersensitivity. Of particular importance is Reye syndrome, a serious and potentially fatal illness, which occurs in children after apparent recovery from viral infections that cause fever. Therefore, the use of aspirin is contraindicated in children.
Administering analgesics
It is easier to suppress a pain at the onset than when it has already set in, so treatment should begin as soon after the painful stimulus as possible. However, some people believe that analgesics should be used only when the patient is in pain (on demand), because in this way analgesic is only used when the patient actually feels pain and prevents the use of drugs during pain-free periods. This practice actually makes it difficult to achieve proper analgesia.
For a correct pain-free condition, which remains steady over time, regular administration (scheduled) is recommended at pre-established intervals, according to the pharmacokinetics of the active ingredient chosen. This way, the possible effects related to overuse would be much less important than the potential pain that the patient would go through unnecessarily (one that persists due to inadequate, insufficient or nonexistent treatment). If one administers the drug when the patient is already beginning to feel pain it means that, between the time the painkiller is requested or administered and the time it takes to reach the plasma levels required to control pain, the patient is not in a pain-free condition and feels pain in the time lapse between doses.
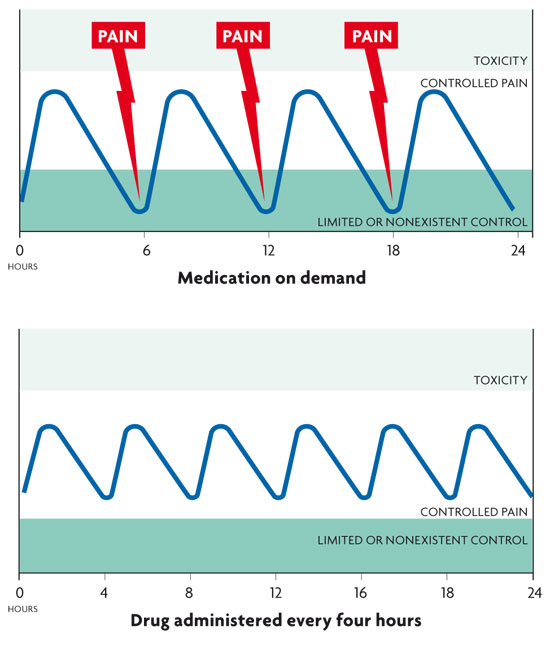
Analgesic efficacy according dose/interval. Administering the drug at the onset of pain means there is a time lapse during which the patient does not get a sufficient analgesic effect (above). When the drug is administered on a regular or scheduled basis, unnecessary pain is avoided (below).
How to use analgesics
According to the 2006 National Health Survey, 62.2% of Spaniards (55% men and 69.2% women) had taken medication in the two weeks prior to the survey. Of these, nearly half, 47.2% (39.1% men and 53.5% women) had taken painkillers, 6.1% for fever and 8.3% for rheumatism, and probably a high percentage of these were NSAIDs or simple analgesics. 71.7% of the medicine taken had been medically prescribed while 28.3% corresponded to self-medication.
According to a report by the Spanish Agency for Medicines and Health Products, in Spain, the use of NSAIDs outside the hospital setting rose in the 1992-2007 period, from 26.30 DDD (defined daily doses, as established by the WHO to compare use of medicines) per 1,000 inhabitants per day in 1992 to 45.81 in 2006. This rise is mainly due to increased use of ibuprofen, which, together with diclofenac and aceclofenac, accounts for almost 70% of consumption.
A comprehensive information study by the Spanish Health System has indicated that at least 20% of the adult population consume NSAIDs daily, but if we take into account that these drugs are easily accessible and many are available over the counter their real use may far exceed the figure mentioned above.
In addition to assessing consumption-related data, one should analyse how they are used. A study assessing the consumption of NSAIDs in Andalusia, and the relationship to prescription and pharmaceutical expenditure, showed consumers were mostly pensioners (66.4%). The authors concluded that the pattern of use was inappropriate and made similar conclusions extending to other Spanish regions.
In Finland, a study of 6,500 patients showed that states of depression and unemployment increased the likelihood of using these drugs, and that the frequent use of analgesics sometimes corresponded to people without justifiable symptoms and also that the joint use of prescribed analgesics and over-the-counter drugs is a risky practice.
An important aspect linked with the consumption of NSAIDs is the simultaneous use of gastroprotective drugs to prevent adverse gastrointestinal reactions. In the nineties, with the advent of proton pump inhibitors (PPIs), these drugs became the therapeutic option of choice for treating gastroduodenal ulcers. In Spain 85 people out of 1,000 are estimated to be taking PPIs daily, while in Norway the corresponding number is only 30 and in Italy it is 27. Accordingly, the use of these drugs is considered very high in our country. In practice, the use of these inhibitors in patients taking NSAIDs is 46-60% of the total prescriptions, which shows that prophylactic use is well over that recommended for treatment.
Abuse of painkillers
The WHO (World Health Organization) defines abuse of analgesics as the consumption of painkillers regardless of medical necessity or in unnecessary quantities. This is subject to various factors, sometimes due to the doctor who, despite prescribing them correctly, does not give a proper explanation of how to take them, or it may depend on the patient, who at the slightest hint of pain, tries to prevent it from developing by taking the painkillers prematurely, or may even become dependent. The ease with which painkillers can be bought also encourages their indiscriminate use.
One of the paradoxical effects of this abuse is chronic daily headaches caused by NSAIDs. This complaint is common in clinical practice and is often directly related to overuse of painkillers, to such an extent it has been defined as a disease in its own right with a prevalence of 1.5%.
Headaches caused by medication overuse are considered as secondary headaches by the International Classification of the Headache Disease, 2006. They can be defined as perpetuation of pain in patients suffering chronic headaches caused by the use of analgesics in excessive amounts and frequencies. The causes of this condition are unknown, although more and more people argue that the main aetiological factor is genetic. Analgesics alone are reported not to cause chronic headaches in patients without a previous record of cephalgia, but they can worsen episodic headaches and thus contribute to chronicity. Any daily or almost daily headache that improves partially (only partially or only for a few hours) on administration of a certain drug, should be suspect to diagnosis as a headache related to analgesic overuse.
In an epidemiological study of headaches caused by medication overuse conducted in Spain, 34.7% misused simple analgesics (most commonly paracetamol/acetaminophen), followed by ergot-containing medications (27.8%), opioids ( 12.5%) and triptans (2.7%), while the rest (27.8%) misused different combinations of drugs.
To treat this type of headache, the Handbook written by the Cephalgia Workgroup of the Sociedad Española de Neurología (Spanish Society of Neurology) recommends discontinuation of the drug, and the treatment of migraine attacks with triptans or NSAIDs. It also recommends treating other factors predisposing to chronic use (sleep disorders, obesity, stress, anxiety or depression) or administering preventive treatment with beta blockers or neuromodulators in the case of migraine, and amitriptyline if there is a tension-associated component from the beginning. Relapses in these patients are common.
In this respect, it is necessary to follow guidelines to use analgesics properly. The right drug must be chosen based on the nature and severity of the pain, using the oral route whenever possible. Appropriate doses must be used at regular intervals (according to the clock and not on demand) and facilitate compliance with slow-release formulations. Finally, secondary or adverse effects should be treated early on. In summary, in the treatment of pain, analgesics should be used according to recommendations and guidelines, and users should be aware of their possible side effects and avoid misuse.
Gastrointestinal reactions:
– Mild gastritis, dyspepsia, heartburn (affecting 15-25% of users). Gastric mucosal erosion, gastric or duodenal ulcers (40% of patients treated for 3 months).
– Bleeding and perforation (less common but more serious).
Renal reactions:
– Reduced renal function in people already affected.
– Retention of water, sodium and potassium (causing hypertension in 3-5% of those treated).
– Chronic renal toxicity (analgesic nephropathy after chronic use).
Hypersensitivity:
– Rhinitis, rash, urticaria, asthma, anaphylactic shock (1-2% of patients).
Hematologic reactions:
– Agranulocytosis, aplastic anemia, thrombocytopenia (rare but potentially very severe).
General adverse reactions of nonsteroidal anti-inflammatory drugs.
NOTE
1. Factors increasing the risk of gastrointestinal complications are previous peptic ulcer, previous gastrointestinal bleeding, over 65 years of age, high doses of NSAIDs, tobacco, alcohol and concomitant therapy with corticosteroids. (Back to text).
BIBLIOGRAPHY
Colás, R. et al., 2004. «Chronic Daily Headache with Analgesic Overuse: Epidemiology and Impact on Quality of Life». Neurology, 62: 1338-1342.
García del Pozo, J. & F. J. de Abajo, 2006. Utilización de antiinflamatorios no esteroides (AINE) en España, 1992-2006. Ministerio de Sanidad y Consumo.
García del Pozo, J., 2009. «Estudio de utilización de antiulcerosos en España (2000-2008)». Información Terapéutica del Sistema Nacional de Salud, 33: 49-54.
Ministerio de Sanidad y Consumo, 2006. Encuesta Nacional de Salud 2006. Ministerio de Sanidad y Consumo.
Romo, C. et al., 1998. «Uso racional de antiinflamatorios no esteroideos en atención primaria». Atención Primaria, 22: 177-186.
Trescot, A. M. et al., 2008. «Opioids in the Management of Chronic Non-cancer Pain: an Update of American Society of the Interventional Pain Physicians (ASIPP) Guidelines». Pain Physician, 11: S5-S62.
Turunen, J. H. et al., 2005. «Frequent Analgesic Use at Population Level: Prevalence and Patterns of Use». Pain, 115: 374-381.