| In recent decades, oncohematology has witnessed major advances in cancer treatment and survival. The toxicity associated with classical chemotherapy, and the need to improve the efficacy and tolerance to treatment, have led to a flood of research activity and many fruitful results. Much of this effort has been focused on developing «targeted therapies» directed against specific functional alterations, which are key to tumour cell survival. Thus, greater treatment efficacy is sought whilst the patient pays a lower cost in terms of toxicity. Advances in other fields of oncology and epidemiology, surgery and radiation therapy have also contributed to the constant advancement of oncology. In short, we glimpse a future ahead that is full of hope for cancer patients and, at the same time, exciting for physicians and researchers alike.
Keywords: targeted therapies, anti-target drugs, tyrosine kinase inhibitors, resistance mechanisms,screening. |
||
|
The magic bullet The major advances in cancer therapy have happened at unprecedented speed. At the beginning of this century, the therapeutic arsenal available to cancer patients has grown considerably, compared to just 20 years ago. In fact, it has been just over a century since, in 1901, the German Nobel laureate Paul Ehrlich enunciated the theory known as the «magic bullet», which referred to the qualities a good antibiotic drug should possess. According to this theory, a particular chemotherapy treatment would be more effective the more it resembled a magic bullet, able to identify and attack only the target for which it was intended, This bullet would leave the remaining healthy tissues intact, reducing treatment toxicity without compromising its effectiveness. At the time, Ehrlich’s hypothesis referred to the first antimicrobial treatments, for example the compound 606, a derivative of arsenic used for syphilis treatment that he himself developed (Calvo, 2006). However, this theory could be applied to other treatments. Specifically, cancer therapy has undergone a process of selection and development of new drugs that are increasingly directed against a specific target, seeking to respect all fully healthy cells. So what Ehrlich baptised in his day as a «magic bullet» is known in the field of oncology today as «targeted therapy» or «molecular target therapy». In fact, if we look back, all the milestones in treatment since the first cytostatics were synthesized in the sixties, they have improved over their predecessors in one of two ways; either granting greater specificity against the tumour, which increases its effectiveness, or is less aggressiveness against tissues and organs, thereby reducing its toxicity. In this sense, the therapeutic options available up until just 15 or 20 years ago were only cytostatic or cytotoxic in nature, producing tumour cell death or arrest, commonly known as «chemotherapy», although this term is inaccurate as any systemic treatment is considered chemotherapy. Cytostatics/cytotoxics form a group of drugs with an arsenal of action mechanisms for inducing apoptosis or cell death by interacting with different systems of the cell, the DNA or the cytoskeleton. The main disadvantage of these drugs is their poor specificity in targeting the tumour cell alone, so their mechanism of action necessarily affects healthy cells. Recently, however, new therapies have been introduced. These are directed against a specific target, like a protein or a membrane receptor in the tumour cell, thus working within a very similar framework to Ehrlich’s «magic bullet». The first anti-target therapies introduced were intravenously administered monoclonal antibodies, however in recent years we have seen a real boost in the development of a new family of orally administered targeted therapies, called small molecule inhibitors. Among them the most common are the tyrosine kinase inhibitors (Arora and Scholar, 2005). The current era: targeted attack During recent years the pace of progress in systemic treatments has reached a truly dizzying pace; obliging professionals to constantly be at the forefront of these changes. In this respect, last year we witnessed many advances both in the field of haematology and oncology, presented at a host of international conferences on these disciplines. For example, we note how the maintenance of chemotherapy with lenalidomide improved the relapse time of multiple myeloma-diagnosed patients, after undergoing hematopoietic cell transplantation. We have also witnessed how the combination of rituximab with bendamustine improved overall survival of patients with mantle cell lymphoma, compared to the classical treatment (R-CHOP). Likewise, treatment of non-hematologic solid tumours has undergone further progress, especially in those therapies directed against key targets in the development and survival of tumour cells. In the area of welfare, a new horizon is opening up, full of new challenges in the treatment of cancer patients. Today novel drugs are already being tested, which could be named hybrids between targeted therapy and cytostatics; these conjugated compounds synthesized in the laboratory combine a monoclonal antibody against a target and a cytostatic attached to the antibody. Thus, the two most advantageous features of each are joined: monoclonal specificity and chemotherapeutic effectiveness in causing cell death or growth arrest. Thanks to this, we can revive familiar chemotherapy treatments, which are highly toxic but very effective, such as DM1 (emtansine), which can be turned into our ally by being attached to an antibody, trastuzumab (T) directed against the Her-2 receptor expressed in some tumour cells in breast cancer. This treatment, known in short as T-DM1, penetrates the cell with high efficiency and minimal toxicity, thus achieving the ideal of every cancer treatment (Roth et al., 2013). |
«In cancer therapy, new drugs have been selected and developed that are directed against specific targets» |
|
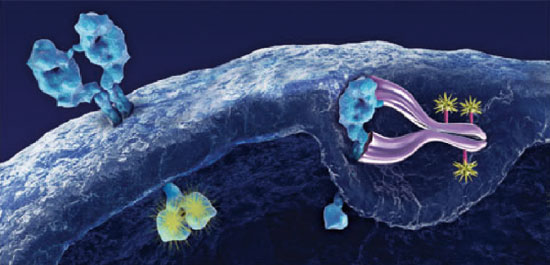 T-DM1, a combined and targeted therapy combining the high efficiency of chemotherapy DM1, with the specificity of trastuzumab (T), an anti Her-2 directed therapy, acting only at the intracellular level in the tumour unit.2013 F. Hoffman-La Roche Ltd. T-DM1, a combined and targeted therapy combining the high efficiency of chemotherapy DM1, with the specificity of trastuzumab (T), an anti Her-2 directed therapy, acting only at the intracellular level in the tumour unit.2013 F. Hoffman-La Roche Ltd. |
«Although cancer treatments are increasingly targeted, over time tumour biology inevitably develops resistance mechanisms towards them» |
|
|
The challenge of winning the fight: importance of exposing the mechanisms Although cancer treatments are increasingly targeted, over time tumour biology inevitably develops resistance mechanisms towards them, which means we must be constantly alert to those alternative signalling pathways that finally lead to treatment failure. Two of the most important examples are found in breast cancer, where new treatments have been discovered for patients who had developed resistance to highly effective drugs such as trastuzumab or hormonal treatment, by inhibiting alternative signalling pathways such as Her-3 (pertuzumab) or the mTOR pathway (everolimus) exploited by the tumour cell for its survival (Roth et al., 2013). Treatment of another highly aggressive tumour, melanoma, has experienced significant changes in recent years, which has gives hope to a significant number of patients. Mutations of another tyrosine kinase, B-Raf, are present in up to 50% of patients with metastatic melanoma, and constitute an essential cell survival mechanism and are the target of the inhibitory treatments of this pathway: Dabrafenib, Vemurafenib or Tramutinib. These treatments have been shown to increase survival in patients, when compared with the conventional and ineffective treatment using cytostatic chemotherapy (Roth et al., 2013). There are other classical mechanisms used to combat tumour progression, for example immunotherapy. Although it has always been a mechanism postulated as desirable, for it would use the body’s own defence system to fight the tumour, rarely has it demonstrated the desired effect. However, we are also seeing significant progress in this field. Thus, in recent years, studies in their initial phases for various tumour types, such as melanoma or lung cancer, have shown that the inhibition of some receptors involved in blocking the cellular immune response against these tumours, such as PD-1 or blocking its ligands, PD-L1, gives encouraging long-term responses in. No doubt, in the near future we will see more advanced studies in this area (Roth et al., 2013). Other important players However, novelty in cancer management is not only evident in the new drugs developed in recent years, but also in newly developed surgical and radiotherapy techniques, as well as the introduction of population screening programmes for early diagnosis. Altogether, they have led to considerable changes in cancer patient management. On the one hand, the development of more sophisticated surgical techniques has contributed significantly to an improvement in disease control and patients’ quality of life. For example, the establishment of conservative surgery for breast cancer has had a fundamental impact on the quality of life of many patients surviving this disease, which can preserve their breasts with reasonable aesthetic results. Moreover, the surgical removal of liver metastases of colon cancer, surgery of the primary tumour in kidney and ovarian cancer, and the removal of lung and brain metastases have a strong impact on patient survival when tumours are in more advanced stages. Other progress has been seen in terms of reduced morbidity or operative mortality, as in the development of less aggressive laparoscopy or gastrectomy. In many cases these innovations have been the result of the introduction of new technology in the operating room. The main consequence in a clinical setting is the increasing need for collaboration between units or even between different hospitals. In line with local treatment, the introduction of new technologies applied to the field of radiotherapy provides more intensive local treatment, with better short and long-term tolerance. Thus, we find many examples of new treatments that have modified previous treatments: new radio-surgery techniques on brain metastases (technique combining minimally invasive surgery and radiotherapy highly targeted and able to hone in on the point of interest), radio-frequency on liver lesions (high frequency ablative vibration) or radio-therapy of modulated intensity. The latter, is a technical mode, which allows an exact concentration of the dose to be applied to the cancerous tumour, while reducing the dose in healthy tissues, by using radiation beams of variable intensity. Furthermore, the introduction of early diagnosis screening programmes and screening in breast cancer or cervical cancer, are responsible for a change in the spectrum of malignancies clinicians face, as they help to detect cancer when in its very early stages. Thus, in these neoplasms in which early diagnosis is implemented a greater number of smaller or less advanced tumours are detected, allowing early treatment and thereby increasing treatment success rates. Also, in the case of colon cancer, recent studies show a decrease in mortality from this tumour due to population screening programmes using the endoscopic study of the distal colon by sigmoidoscopy. Retrospective studies have shown that, since the eighties, the combination of new therapies, more sophisticated surgical techniques and the introduction of population screening programmes combined, have had a significant impact on reducing cancer mortality. In fact, it is estimated that between 28% and 65% of the decline in mortality is attributed to the benefit of adjuvant therapy and early diagnosis in breast cancer (Berry et al., 2005). |
«New surgical and radio-therapy techniques and the introduction of population screening programmes have had a significant impact on cancer-patient management» |
|
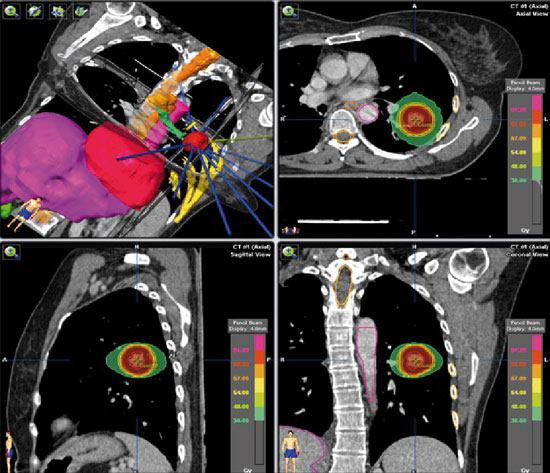 The new increasingly directed techniques of radiation therapy, such as stereotactic radiotherapy, can improve treatment and reduce its toxicity. Above, high gradient dosimetry of a extra-cranial stereotactic radiotherapy treatment of a pulmonary nodule.Source: Fernández-Velilla Peña et al., 2013. The new increasingly directed techniques of radiation therapy, such as stereotactic radiotherapy, can improve treatment and reduce its toxicity. Above, high gradient dosimetry of a extra-cranial stereotactic radiotherapy treatment of a pulmonary nodule.Source: Fernández-Velilla Peña et al., 2013. |
«Collaboration between clinical and preclinical research, namely translational oncology, will be the cornerstone of future research endeavours» |
|
|
Certainly, the history of cancer management is full of exciting developments and therapies, which have represented a real challenge for clinicians, who have had to keep an open mind to change and remain in a state of constant learning. In the future we will have to apply what we have learned and continue to innovate while maintaining the ways of working that have brought us so much success in the past. In this sense, looming on the horizon are new challenges, for both the researcher and the clinician. First, as mentioned, preventive mass screening or early diagnosis has meant a change in the spectrum of tumours, significantly increasing cancer diagnosis in its early stages (Pass et al., 2004). This is a huge advantage for the patient, who is offered treatment within a curative strategy. But many questions still remain unanswered in this field. For instance, the recommended age for starting and stopping screening tests is still under debate. Regarding breast cancer, the benefits of starting early diagnostic screening at 40 years of age are debatable, and nor is there enough information on whether elderly patients (over 75 years) benefit either. This debate about the age range or the frequency of screening tests is applicable to other programmes to screen for other tumour types. Also, the emergence of new imaging and laboratory techniques opens up new untapped possibilities for application to other cancer screening programmes. Finally, new therapies have brought real changes to cancer treatment; however, there is still a long way to go and it seems that the more progress we have seen in the past, the more progress we demand for the future. Therefore, in this context, there is a strong need for clinical research and development of new compounds, which will doubtless be a journey full of obstacles and difficulties. Again, we can quote the visionary Paul Ehrlich. Medical research for this German doctor was supported by four pillars, which he called the four Gs: Geld («money»), Geduld («patience»), Geschick («intelligence») and Glück («luck»), representing problems and needs still vividly relevant today. In the field of cancer research, close collaboration has been essential between clinical and preclinical studies to achieve many past successes. In fact, a new term has been coined, known as «translational oncology», which defines the interaction between clinical, basic and epidemiological research as a means to reduce cancer incidence and mortality. The development of targeted therapies and improved knowledge of new molecular and genetic predictors may be considered as two examples in which the interaction between clinical and preclinical research has achieved satisfactory results. Great progress is being made in this respect, as evidenced by the growing number of communications in this field at major international conferences. Platforms investigating genomic profiles that allow procurement of information about both tumour biology and the effectiveness of treatments are essential. Projects such as the Cancer Genome Atlas and the Cancer Cell Line Encyclopaedia provide us with invaluable information about many cell lines and extensive intratumoral heterogeneity. Therefore, in the future, collaboration between clinical and preclinical research, i.e., translational oncology, will be the cornerstone of upcoming research endeavours. Thus, there is an increasing demand for clinicians to increase their knowledge in molecular biology, and be increasingly involved in research. Likewise, pre-clinical researchers are expected to coordinate themselves with the medical staff in hospitals, to take their discoveries from the laboratory bench to the patient’s bedside. Furthermore, regarding drugs with specific molecular targets, we are witness to the ever-increasing development of a plethora of compounds. They act against various proteins and receptors present, not only in the tumour cell membrane, but also in the cytoplasm and nucleus, achieving the alteration and sometimes even the arrest of signalling pathways essential to tumour survival. So, these new drugs against signal transduction pathways or against enzymes, involved in DNA replication or mitosis, are starting to show positive results in phase I studies, and are expected to make the leap to the patient’s bedside in the years to come. Ultimately, ahead of us lies a future full of hope for cancer patients, while exciting for physicians and researchers alike. In fact, taking into account the relatively short period of time in which huge leaps in cancer treatment have been taken, in the relatively near future we expect to see a significant growth in therapeutic options and, thus, an obvious improvement in patient survival. |
||
REFERENCES
Arora, A. and E. M. Scholar, 2005. «Role of Tyrosine Kinase Inhibitors in Cancer Therapy». The Journal of Pharmacology and Experimental Therapeutics, 315: 971-979. DOI: <10.1124/jpet.105.084145>.
Berry, D. et al., 2005. «Effect of Screening and Adjuvant Therapy on Mortality from Breast Cancer». The New England Journal of Medicine, 353: 1.784-1.792. DOI: <10.1056/NEJMoa050518>.
Calvo, A., 2006. «Ehrlich y el concepto de “bala mágica”». Revista Española de Quimioterapia, 19: 90-92.
Fernández-Velilla Peña, M. et al., 2013. «Tratamiento de lesiones pulmonares mediante radioterapia estereotáxica extracraneal tras colocación guiada por tomografía computarizada de marcador Viscoil®: experiencia inicial». Radiología, 55(3): 225-232. DOI: <10.1016/j.rx.2011.09.020>.
Pass, H. et al., 2004. «Changes in Management Techniques and Patterns of Disease Recurrence over Time in Patients with Breast Carcinoma Treated with Breast-conserving Therapy at a Single Institution». Cancer, 101(4): 713-720. DOI: <10.1002/cncr.20410>.
Roth, B. J. et al., 2013. «Clinical Cancer Advances in 2012: Annual Report on Progress Against Cancer from the American Society of Clinical Oncology». Journal of Clinical Oncology, 31(1): 131-161. DOI: <10.1200/JCO.2012.47.1938>.





