
|
A long time has passed since 1683, when the Dutch naturalist Anton van Leeuwenhoek observed spermatozoa for the first time using a homemade microscope. He called them «animalcules». Science progressively revealed those diminutive «animalcules» were responsible for human existence itself. Later, the German physician Robert Koch would discover bacteria in the bloodstream of anthrax-infected cows. The idea that bacteria caused disease was firmly accepted. That is, after a long period of scepticism. Soon scientists described the characteristics of microorganisms, classified them, and found the way to take advantage of such information for the benefit of humankind. In fact, the use of microorganisms to help people has existed since the old times, in a completely unconscious and empirical way. There are countless applications for microorganisms, namely in the process of obtaining food, as is the case with fermented products: wine, beer, yoghurt or bread. In the medical field there have also been great advances, like fungus-produced antibiotics, the greatest breakthrough in medicine that is Alexander Fleming’s discovery of penicillin in 1928 or Edward Jenner’s vaccines in 1796. It may seem – a priori – that humans have managed to control microorganisms, but that is not the case at all. Despite the great scientific advances in antibiotherapy, microbiology and epidemiology, which allowed us to eradicate high-morbidity viruses such as smallpox, an important part of microorganisms are still responsible for pandemic diseases for which we still do not have a treatment. So much so that certain infectious agents have been studied as possible biological weapons to be used in wars or in bioterrorism, and there are other that have not been controlled or eradicated yet and represent the biggest challenge in the twenty-first century, such as AIDS (Acquired Immunodeficiency Syndrome). There is a group of globally relevant infectious diseases called zoonoses. A zoonosis is an infectious or parasitic disease transmitted from animals to people. Currently, according to the OIE (World Organisation for Animal Health), 60% of human pathogens are zoonoses and 75% of them are emerging. An emerging disease is one that was previously unknown, or was known but did not represent an important problem before. The Q Fever, a 21st Century Zoonosis An example of emerging zoonosis is the Q fever. The disease was first described in 1935 in Australia by Edward Holbrook Derrick, a doctor in Queensland who described an outbreak of the disease in a group of workers of a cattle slaughterhouse. A year later, after several frustrated attempts of associating the outbreak with a known microorganism, Derrick sent samples from his patients to a colleague, Frank Macfarlane Burnet, a Melbourne doctor. He was the one to isolate the previously unknown bacterium. Concurrently, in the United States in 1937, Herald R. Cox isolated the same microorganism from ticks. The name of Q fever was coined by its first discoverer, Derrick. «Fever» was the main symptom and «Q» came from “query” and referred to the unknown cause of febrile cases. Regarding the infectious agent, the responsible bacterium, it was finally named Coxiella burnetii to honour doctors Cox and Burnet. Due to its characteristics (high environmental resistance, high infectivity, airborne transmission and high infectious capacity), in the forties, during World War II, the bacterium was used in many experiments and trials in order to turn it into a biological weapon by countries such as the old Soviet Union, the United States of America and Japan. However, the disease was not really seen as a health threat until the first decade of the twenty-first century, when a number of Q fever cases emerged in several countries of the European Union. Surely, the most remarkable outbreak was the one to surface in the Netherlands from 2007 to 2010. In 2009, there were 2,357 human cases, six of which were fatal. Furthermore, the livestock industry, specially goat farmers, suffered great economic losses due to abortions. As of that moment, the Q fever was considered an emerging zoonosis, and has been the object of many investigations to this day, because its pathogenesis is still a mystery. What Do We Know about the Q Fever Today? We currently know that Coxiella burnetii is the bacterium causing the Q fever. It is an obligate intracellular, i.e. lives inside the cells of the host. The disease is present almost everywhere in the world and has been isolated from a large number of hosts, from invertebrate organisms such as ticks to more complex organisms such as wild animals, domestic mammals and, obviously, humans, too. The main reservoirs and sources of infection for humans are domestic ruminants, namely sheep, goats and cows. Infected animals can excrete the bacterium to the environment through milk, faeces, vaginal fluid, urine, semen and the product of abortions and births (fluids and placenta). The moment when the excretion is maximised is in the period around labour or abortion, although it is also possible in any other moment. The bacterium forms contaminated aerosols from these materials and travels airborne, helped by the wind, covering great distances until it is inhaled by a susceptible individual. This manner of transmission helps the infection escape the more restrictive circle of people who have direct contact with animals, like livestock breeders or veterinarians, and infect people far from that environment. Once inhaled, the bacteria reach the lungs of the new host and are absorbed by the immune system cells. They spread from those to the rest of the body, to be finally excreted again into the environment, thus closing its life cycle. The disease pattern of Coxiella burnetii in humans has been extensively described. It has been estimated that in 60% of the cases, the infection can go unnoticed and present no symptoms (subclinical infections). Nonetheless, when it does show, the symptoms are similar to those of a flu case, with a fever and respiratory problems. In chronic cases, and specially in old or immunodeficient patients, more serious diseases such as hepatitis or endocarditis can appear. In pregnant women, abortions have also been described. In fact, it is commonly believed that most of the abortions of unknown origin are due to this bacterium. |
 Tim Ertl Tim ErtlIn domestic mammals, main reservoir of the bacterium, the disease has not been studied too much, and different studies reach conflicting results. In general terms, the bacterium is said to cause reproductive issues such as abortions, infertility and preterm labour, among others. «Despite the great scientific advances, an important part of microorganisms are still responsible for pandemic diseases for which we still do not have a treatment» |
|
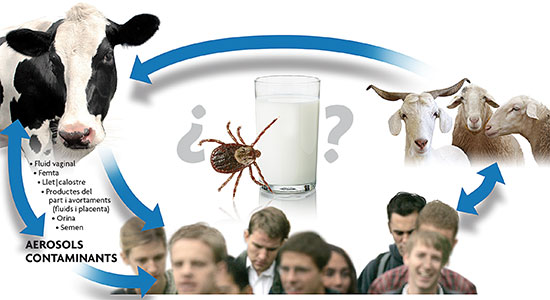 Mètode MètodeSchematic representation of the life cycle of Coxiella burnetii. In the picture, the epidemiological role of transmission through the intake of contaminated raw milk or due to ticks as a possible transmission vector. |
«The disease was not really seen as a health threat until the first decade of the twenty-first century, when a number of Q fever cases emerged in several countries of the European Union» |
|
|
What Do We Not Know about the Q Fever Today? In domestic mammals, the main reservoir of the bacterium, the disease has not been studied too much, and different studies reach conflicting results. In general terms, the bacterium is said to cause reproductive issues such as abortions, infertility, preterm labour leading to weak or dead offspring, endometritis, placentitis, placenta retention at birth or even mastitis. All these symptoms point to the Q fever becoming a serious public health problem. Besides, it can also represent a serious economic problem for sheep, goat and cow farmers, very important economic sectors in Spain. It is very necessary, therefore, to make the study of how the disease affects domestic ruminants one of the main focuses of research on the Q fever. If we manage to control the disease in cattle, the risk of infection for human populations will also decrease. Currently, bovines are the domestic ruminants that raise more doubt. Maybe because there are more hard to detect subclinical infections in the species, or because we still do not understand the mechanisms through which the bacteria harm the host. In order to control the disease, it is essential to have enough tools to diagnose it. This is currently a very difficult clinical task. On the one hand, the disease is subclinical in a majority of animals. On the other, symptoms, when present, are non-specific. Therefore, it is essential to have the aid of laboratory techniques to confirm the diagnosis. Serological techniques, based on the detection of anti C. burnetii antibodies in blood plasma, and molecular biology techniques, based on the detection and amplification of bacterial DNA fragments, have been the most successful ones, but they have to be combined to be certain of the diagnosis, since there are seropositive animals that do not excrete the bacteria and vice versa. The question we still need to answer today is whether or not these differences in serology and excretion are meaningful or depend on individual variation. Can the Q Fever be Treated in Animals? In human medicine, the chosen treatment is tetracycline, an antibiotic, for a long period. In animals, however, antibiotherapy is restricted due to three evident reasons: 1) there must be a suppression period, that is, milk and meat cannot be marketed until some days after the therapy, 2) the cost of the treatment is too high, and 3) antibiotherapy for this disease has not proved very effective in animals. Control of the Q fever in livestock with prevention measures involves a series of procedures to try to reduce the possibility of contagion from one cow to another or to a person, specially to risk subjects. The main methods are: keeping the facilities in good hygienic conditions and with good ventilation, specially the areas where the animals are; removing the placenta in births and abortions as soon as possible, always handling it with gloves; avoiding manure removal during the windy days from the facilities where are the animals are housed, as the bacteria will be dispersed more easily during those days; keeping wild animals, as well as dogs or cats, from entering the location, enclosing the perimeter of the holding; pasteurising or sterilising the milk if it will be consumed by humans; and, of course, vaccinating the animals against Coxiella burnetii in order to immunise them. |
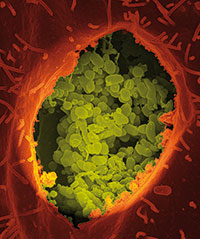 National Institutes of Health (NIH) We currently know Coxiella burnetii is the bacterium causing the Q fever. It lives within the cells on the host. The disease is present almost everywhere around the world and has been isolated from a large number of hosts: from invertebrate organisms, such as ticks, to superior organisms, like wild animals, domestic mammals and, of course, people, too. In the picture, dry fracture of a Vero cell exposing the contents of a vacuole where Coxiella burnettii grows. «The Q fever can also represent a serious economic problem for sheep, goat and cow farmers, very important economic sectors in Spain» |
|
 National Archives of Australia: A1200, L36826 (esquerra) / U.S: National Library of Medicine (dreta) National Archives of Australia: A1200, L36826 (esquerra) / U.S: National Library of Medicine (dreta)The infectious agent, the bacterium causing the disease, was named Coxiella burnetii after its discoverers, Cox and Burnet. On the left, Frank Macfarlane Burnet. On the right, Herald R. Cox, working in the laboratory. |
«Although the Q fever has been problematic at different moments throughout history, it looks like a “benign” process for now» | |
|
The Q Fever in Bovines, the Controversy Coxiella burnetii, the agent responsible for the Q fever, is a bacterium that is present in the environment and cows from dairy farms. Its presence in these hosts is undeniable and is confirmed by the presence of specific antibodies in the animals, and also the DNA detected in samples from different sources such as vaginal fluid, milk, faeces and placenta. However, the possible connection of the bacterium with reproductive problems described in different works and papers is not clear at all – it is actually quite debated. There are those who defend the idea that the bacterium causes clinical symptomatology in a number of animals. The other opinion is that the infection is subclinical and represents no problem for livestock facilities. A possible explanation for this chaotic scenario is that the least productive animals and those with reproductive problems – specially those with an infertility record – are eliminated from farms, because it is economically harmful for the farmer to keep those animals in the farm. A different justification is based on the strain. Within the same bacterial species, strain designates a group of bacteria that descend from a cell and are, therefore, genetically identical. So within the same bacterial species we can fin specimens with different characteristics pertaining to different strains. This makes it possible for branches of Coxiella burnetii with different virulence to exist. We understand virulence as the ability of bacteria to damage their host. Luckily, it looks like the currently dominant strains in Spain are not too virulent. This fact would also explain how difficult it is to link infection and clinical disease. Probably both theories are true and we are just observing the joint effect of both phenomena, the effects of removing unproductive animals from herds and the presence of low-virulence strains. Q Fever, Let’s Be Realistic It is easy to reach such conclusions. Especially since a reassuring and comfortable message can be extracted for society in general. We could, therefore, conclude that although the Q fever has been problematic at different moments throughout history, it looks like a «benign» process for now. It is there, but it is not harmful. However, that would be so short-sighted. Mutations are permanent DNA changes that are transmitted to the offspring. Bacteria experiment this phenomenon that makes it possible for new bacterial strains to appear. The changing state of the strains due to mutation answers the question: why has the Q fever been the cause of large disease outbreaks, but now it seems not to be a threat? It is a feasible hypothesis, because it is a common situation for different infectious agents, such as the flu virus. Therefore, it is necessary for research not to stop, to advance diagnostic methods so they are more efficient and inexpensive (not only for research but also to better fight infection in livestock locations) and to improve control of the livestock movements in order to minimise the risk of introducing a virulent strain in a new area. Now we are living good times, but virulent strains can appear any moment and we need to be prepared and move ahead of the events. The Q fever, which seems a false alarm now, is a real threat. |
«If we manage to control the disease in cattle, the risk of infection for human populations will also decrease» | |
This article received the latest Joan Lluís Vives Scientific Communication Award from the Vives University Network.





