|
«Even being born results in a loud cry», Raimon sings. There are two crucial moments that demarcate human existence: birth and death. Death comes in many forms, agonising or peaceful, with slow suffering or sudden trauma, while we are conscious and when we are not. Birth, however, is almost always accompanied by a loud cry: it is a cry we need to breathe independently and a reaction to the shock of being expelled from the comfortable uterus of our mother out into a hostile, dry and cold environment, which we will not abandon until the day we die. Both our life and our death are accompanied by microbes. Over the course of our existence, we are isolated from contact and interaction with microorganisms only during the first nine months (in utero), or so we thought hitherto. Already during the delivery process, our skin and digestive tract become colonised. As the amniotic sac ruptures, the placenta detaches which frees us to pass through our mother’s vagina and results in the «contamination» of our delicate skin with microbes. We continue to incorporate microbes immediately thereafter, beginning with the inside of our mouths and the rest of our digestive tract, due to contact with our mother’s body and with the environment. During the course of a regular and healthy pregnancy, the mother ‘s body undergoes important anatomical, physiological and biochemical changes. First, there is an increase of body fat; then, as pregnancy progresses, there is a reduction in insulin sensitivity. During pregnancy, there are elevated levels of circulating cytokines, resulting in an increase in inflammation and a metabolic syndrome leading to a condition similar to obesity. This physiological «obesity» and the loss of insulin sensitivity are beneficial, as they favour foetal growth and prepare the mother’s body for the energy demands of lactation. Also during pregnancy, there is a change in the maternal immune system to prevent rejection of the foetus or the placenta. On the other hand, the inflammatory response of the mucous membranes increases, which can lead, for instance, to gingivitis. The placenta allows oxygen and nutrients to reach the foetus and waste products to be removed, without the mixing of maternal and foetal blood. Therefore, the placenta intervenes in nutrition and growth control and regulates metabolism, fulfilling the functions of the foetal intestines, lungs and kidneys, which only function autonomously beginning at birth. Major changes in the intestinal and vaginal microbiota of the mother during pregnancy have been described. But there are also important changes in the microbiota of both the maternal oral cavity and skin and the placenta. These changes in the maternal microbiota are essential for the future development of a «healthy» microbiota in the newborn. Pregnancy is accompanied by a decrease in the microbial diversity of the vagina, as Lactobacillus species such as L. crispatus, L. jensenii or L. johnsonii become dominant. The prosperity of these species is of biological significance. For example, L. jensenii metabolises glycogen, whose presence increases with a rise in oestrogen levels and contributes to vaginal acidity. Furthermore, L. jensenii can present proteins on the cell surface that inhibit certain pathogens causing sexually transmitted diseases, such as Neisseria gonorrhoeae. Finally, L. crispatus and L. jensenii produce hydrogen peroxide, which protects the vagina from certain bacterial infections. After birth, L. johnsonii colonises the intestinal tract of the newborn baby and helps it to digest milk. |
«The placenta fulfills the functions of the foetal intestines, lungs and kidneys, which only function autonomously beginning at birth» |
|
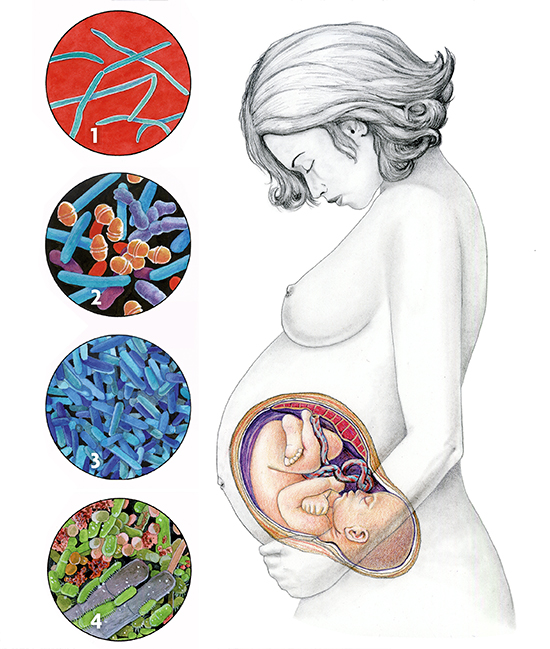 Microbiota of different parts of a woman’s body. 1. Fusobacterium in amniotic fluid (only in pathological situations, in women who suffer preterm labour). 2. Normal microbiota of the placenta, represented by a low number of bacteria pertaining to different groups (such as Firmicutes, Tenericutes, Proteobacteria, Bacteroidetes and Fusobacteria). 3. Microbiota of the vagina, where Lactobacillus acidophilus usually predominates. During pregnancy, other species of the same genus, such as L. crispatus, L. jensenii and L. johnsonii are abundant. 4. Microbiota of the skin of a moist area, such as the pubis, where the phyla Firmicutes and Actinobacteria predominate. / Illustration: Carles Puche
|
||
|
The long-held notion that the placenta is free of microbes has been recently challenged by observations suggesting that it harbours both gram-positive and gram-negative microorganisms. Generally, these microorganisms are not pathogenic. However, in some cases, their composition can vary in response to poorly understood pregnancy-related disorders, such as preterm labour, which occurs in one out of ten pregnancies. Inflammation of the placenta can cause premature contractions and preterm labour. These findings reinforce previous results linking maternal periodontal disease with preterm labour. The foetal intestinal tract was also considered to be microbe-free but exposure of the foetus to microorganisms might actually start before birth, with microorganisms that colonise the placenta. The ability of pathogenic microorganisms, such as the bacteria Treponema pallidum (syphilis) and Listeria monocytogenes (listeriosis) or the HIV virus (AIDS), to cross the placental barrier and enter the body of the foetus through the blood has long been recognised. Congenital infection with these and other pathogenic microorganisms may have serious consequences for the newborn and can even lead to death. The bacterial composition of the intestinal tract of a newborn only a week after being born is already complex, dynamic and fluctuating. A balanced microbiota similar to that of adults does not develop in children until around the end of the first year of life, although in the first week of life, the intestine is already colonised by Actinobacteria (including Bifidobacterium), Proteobacteria, Bacteroides and, to a lesser extent, Firmicutes (or gram-positives, such as Lactobacillus, which is the predominating microorganism in the vagina). Conversely, in preterm babies under 1,200 grams, Firmicutes and Tenericutes predominate whereas the presence of Actinobacteria is reduced. After birth, the choice between maternal or artificial lactation strongly impacts the child’s microbiota. Maternal lactation selects for those microorganisms that use the constituents of breast milk; for example, the oligosaccharides in human milk are the main growth factors for Bifidobacterium and Bacteroidetes. The history of microbiology can be divided into three golden ages. The first is, surely, the one started by Pasteur and Koch during the last quarter of the nineteenth century, when most infectious diseases were identified. The second corresponds to the 1940s and 1950s, when the discovery and application of antibiotics became the first effective weapon against infections. The third one occurred during the last two decades of the twentieth century, as the age of genomics. But now, in the twenty-first century, we are entering a fourth golden age, in which the sequencing of all the microorganisms in a particular location (skin, mouth, intestine, etc.), or metagenomics, has made it possible to discover the presence and possible function of microorganisms whose existence we never even suspected. Indeed, in the healthy mouth, there are an estimated 700 species of bacteria—although the usual concept of species is different in bacteria than in animals and plants. In recent years there have been great advances not only in the identification, isolation and culture of the microbial species in the intestinal tract, but also in the development of genetic tools and models to study the interaction between the microbiome (the set of genes of the microbes in our body) and the genetics and physiology of the host. These and other, yet to be developed tools will be used to perform new studies, thereby increasing our comprehension of molecular targets in the homoeostatic interactions between the intestinal microbiota and our body. This knowledge will reveal new treatments for diseases and thus improve health. |
Search
© Mètode 2014 - 83. Online only. The Digits of Science - Autumn 2014
Ricard Guerrero
Member of the Institute of Catalan Studies and Academic Director of the Barcelona Knowledge Hub of the Academia Europaea.
Mercè Berlanga
Tenure-track 2 professor of the Department of Microbiology and Parasitology of the University of Barcelona (Spain).





