Plants on demand
Genome editing for plant improvement

The plants we eat are the outcome of a humans’ long history of domestication of wild species. The introduction of CRISPR/Cas gene-editing technology has provided a new approach to crop improvement and offers an interesting range of possibilities for obtaining varieties with new and healthier characteristics. The technology is based on two fundamental pillars: on the one hand, knowing complete genome sequences, and on the other, identifying gene functions. In less than a decade, the prospect of being able to design plants on demand is now no longer a dream, but a real possibility.
Keywords: crops, plant breeding, CRISPR/Cas9, genome editing.
The plants we eat
Plants are essential components of terrestrial ecosystems and are indispensable for maintaining life on Earth. The kingdom Plantae includes an enormous range of groups: bryophytes (mosses and liverworts), pteridophytes (ferns and horsetails), gymnosperms, and the extensive group of flowering plants known as angiosperms (monocotyledons and dicotyledons). The highest diversity and, quite possibly, the largest number of unidentified species concentrates in the tropical areas of our planet.
«Despite the apparent variety of plants used in human diet, only around 200 are consumed worldwide on a regular basis»
It is estimated that there are between 450,000 and 500,000 different species of known plants,1 more than half of which are edible. Despite the apparent variety of plants used in human diets, only around 200 are consumed worldwide on a regular basis. In addition, three plants – maize, rice, and wheat – provide almost 60 % of the plant-based proteins and calories in human diets (FAO, 1999). The loss of agricultural diversity has become a growing concern over the last hundred years and is associated with changes in farming practices and the globalisation of production systems. The result is the replacement of multiple local varieties with a small number of new and improved or exotic varieties.
Today’s most pressing agricultural challenges are eradicating hunger and malnutrition and ensuring the growth of enough food to meet the demand of present and future generations (FAO, 1983). The FAO’s global food insecurity estimates from 2012 showed that economic growth was particularly effective in reducing hunger and malnutrition. On the other hand, such economic growth must also take nutrition into account and ensure both the diversification of human diets and the intake of adequate nutrients, all within sustainable production and consumption systems (FAO et al., 2012).
In recent years, the prevalence of food allergies and their global effect on the world’s population have become the subject of analysis. Food sensitivities can be classified as allergies or intolerances; the former involves abnormal immune reactions to food components while the latter occurs in individuals that are sensitive to particular foods but who do not present an immune response to them. Some staple foods in the human diet such as gluten-containing cereals (wheat, barley, rye, oats, spelt, and hybrid varieties or by-products), soy, and nuts like peanuts and walnuts are among the most common plant-based product sensitivities. In particular, gluten intolerance affects 1 % percent of the European population. The recommended treatment for these allergies or intolerances is to remove the particular food item or additive that causes the reaction from the diet. In this sense, the food industry must provide more extensive information on their products’ labels (Taylor & Hefle, 2001).
Innovation in plant breeding
The improvement of plant species for human consumption was an ancestral practice that resulted in the creation of plants with new traits (Figure 1). It was used to obtain higher quality, more productive varieties, or plants that were more resistant to adverse environmental conditions and pathogens. In addition, plant breeding projects have also tried to obtain plant varieties with completely new traits that were not present in the original species (e.g., different plant or fruit colours or morphologies, the absence of seeds, etc.). The primary source of genetic variability for conventional plant breeding techniques are the random mutations generated during the processes of mitosis or meiosis, or during induced mutagenesis (using chemical compounds or irradiation to induce mutations).
«The improvement of plant species is an ancestral practice that resulted in the creation of plants with new traits»
In many cases, the mutation associated with the desired trait must be incorporated into another plant by hybridisation and then selected in the new population resulting from the cross. In this way, one or a few genes are transferred from one variety or donor species into another (the recipient is known as the recurrent parent). Several generations of offspring also crossed with the recurrent parent are required to select the presence of the desired trait in the next generation. This process has limitations related to the precision of the genome fragments introduced into the recurrent plant and the introduction of the desired trait requires long periods of time. However, the development of next-generation sequencing methods means that the complete genomes of many crops are now known. This information allows the combination of conventional breeding technologies with molecular tools which can accelerate the selection process in the generated materials.
The emergence of genetic engineering techniques led to a substantial leap in the ability to generate new genetically modified plant varieties (GMOs). Over the last thirty years, the work of many laboratories has led to the development of a wide variety of plants with interesting modifications; however, although their introduction onto the market was very important, only a dozen different crops have reached a commercial stage (Beltrán, 2018). The two main lines of crop improvement have focused on obtaining herbicide-tolerant and insect-resistant plants.
One of the main reasons why very few modified varieties are on the market is the high cost for the licences required to authorise new varieties. Furthermore, the social perception – mainly in Europe – that GM foods could pose a threat to health has blocked their use, as well as the emergence of new varieties. However, an exhaustive review of the scientific studies published in recent years found no evidence that varieties obtained through genetic engineering pose a higher risk to health or the environment than those obtained through conventional improvement processes (National Academies of Sciences & Medicine, 2016).
Genetic editing in crop plants: innovation and breeding
A new technology which allows genes to accurately be edited has now been available for around six years. CRISPR/Cas2 technology makes it possible to improve plant varieties without introducing foreign elements into them. In particular, the CRISPR/Cas9 system has enormous potential, thanks to its simplicity (Figure 2), low cost, and applicability to a large number of species.
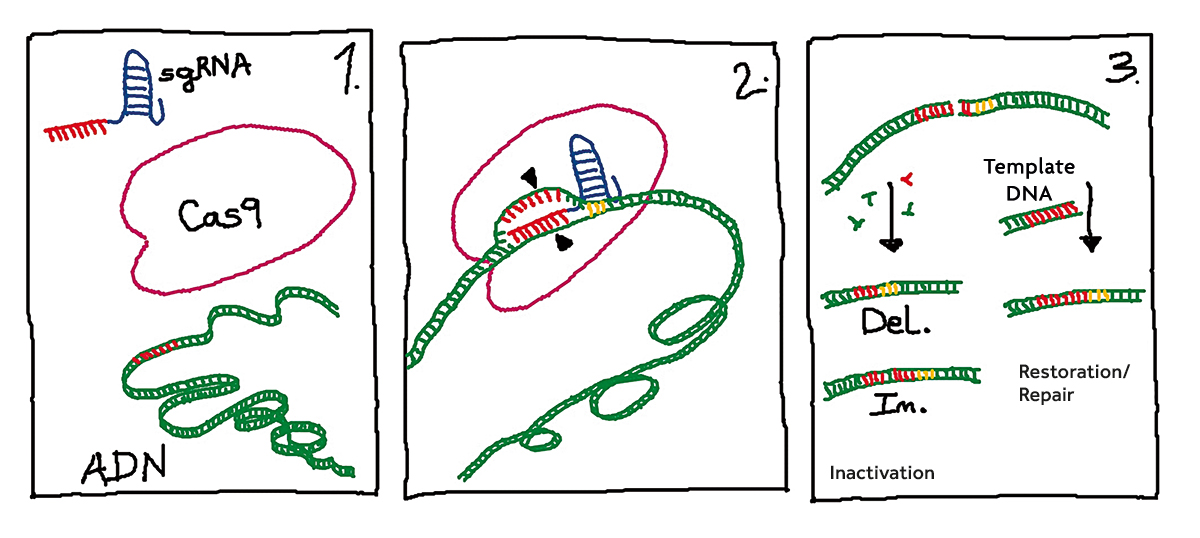
Figure 2. Basic gene editing stages using CRISPR/Cas9 technology. In stage 1, the edition elements (sgRNA and the Cas9 protein) are introduced into the target cells containing the genomic material (DNA). The sgRNA carries a 20-nucleotide guide sequence (in red) complementary to the target gene. In stage 2, the sgRNA/Cas9 complex interacts with genomic DNA and the pairing between the guide sequence from the sgRNA and the target gene occurs. The Cas9 protein recognises a sequence of three nucleotides known as PAM (in yellow) and cuts the double-stranded DNA. In stage 3, the cellular repair mechanisms are activated. On the left, the break repair can only occur at the expense of introducing (In.: inserting) or losing (Del.: deleting) nucleotides, which usually leads to gene inactivation. The right side shows the homologous recombination repair mechanism which requires a DNA template to restore the original sequence or to introduce our chosen change. / Concha Gómez Mena
The discovery of the CRISPR/Cas systems goes back to studies of microbial adaptive immunity systems (Mojica & Montoliu, 2016). Some microorganisms, mainly archaea, protect themselves against viruses by degrading their genetic material. Fragments of this external genetic material are incorporated into their own bacterial genome as a reminder of the invasion. These fragments can guide nucleases (Cas proteins) during future infections to help stop and inactivate the invading viruses. Eric Lander (2016) published a detailed summary of the main researchers involved in the discovery and the study of the molecular mechanisms of the system.
The development of CRISPR/Cas9 technology for eukaryote gene editing is based on the in vitro reconstitution of three elements identified in Streptococcus pyogenes: two RNAs (crRNA and tracrRNA) and the Cas9 protein. CrRNAs (CRISPR RNAs) contain sequences homologous to the target genes present in the genome which are subjected to editing, while tracrRNAs function as an anchor for the Cas9 protein. Cas9 is an endonuclease that cuts double-stranded DNA. The only requirement for the cut is the presence of a three-nucleotide sequence called PAM3 in the region adjacent to the target (Mojica et al., 2009). The double-strand break is repaired by the cellular machinery by randomly removing and introducing nucleotides and thus generating new pairing locations between both strands. If the repair occurs near a gene’s coding sequence, the changes usually result in the inactivation of that gene (Figure 2). A second mechanism allows the double-strand break to be repaired, in a process of homologous recombination (Figure 2). This second pathway makes it possible to repair specific sequences and might have applications in biomedical science as a gene therapy strategy for the treatment of congenital diseases (Montoliu, 2019).
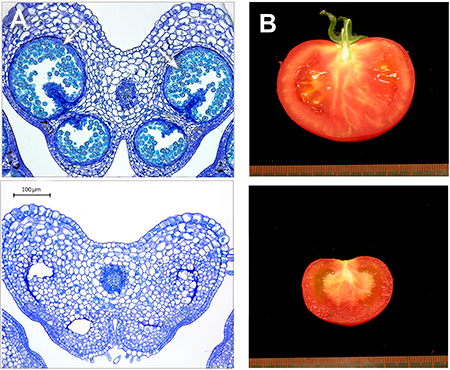
Figure 3. Obtaining pollen-free tomato plants using CRISPR/Cas9 technology. In the edited plant (bottom), a nucleotide has been inserted into a tomato gene sequence necessary for the process of male gametogenesis. A) Histological sections of anthers from tomato plants. Pollen (arrows) can be observed in conventional plants (top), while the edited plants (bottom) are sterile and do not produce it. B) Ripe tomato fruits from the conventional Moneymaker variety (top) and an edited plant (bottom). The edited plants are parthenocarpic – they grow seedless fruit. / Photos by Mª Jesús López Martín
In 2012, the work of two laboratories proposed fusing both ARNs into a single molecule called sgRNA (single-guide RNA) and they proved that this new molecule was still functional and could direct the Cas9 protein to the genome area containing the target (Gasiunas et al., 2012; Jinek et al., 2012). The authors also predicted the potential of this system as a tool for programmable nuclease-mediated genetic editing (Jinek et al., 2012). Shortly after this, the first publications reporting in vivo gene editing appeared in the scientific literature. In 2013, the first papers describing gene editing in mammalian cells (Cong et al., 2013) and plants (Shan et al., 2013) were published.
Since then, the use of this technology to modify the genes of different organisms has exponentially grown. Different studies have proved that it is possible to edit more than one location of the genome simultaneously by using a single nuclease and several sgRNAs, which is especially interesting when we want to edit large genes or gene families (Wolter et al., 2019). Gene editing in plants often requires an initial stage of genetic transformation to introduce the elements of the system (sgRNA and the Cas9 protein) followed by their subsequent removal in the next generation. This is a limitation for species in which efficient genetic transformation protocols have not been developed or those with long generation times, such as some woody plants. In this sense, alternative methods using protoplasts or immature embryos, that do not require a stable transformation are being developed (Liang et al., 2017; Osakabe et al., 2018). Editing systems solely comprising protein or RNA-protein complexes have also been developed to edit genomes without using recombinant DNA (Metje-Sprink et al., 2018).
The future of food and genetic editing in plants
Regarding crop plants, the CRISPR/Cas9 system has been used to improve cereals such as maize, rice, and wheat, horticultural varieties such as tomato, potato, cucumber, watermelon, and woody species including citrus, papaya, and apple trees (reviewed by Wang et al., 2019; Wolter et al., 2019).
CRISPR/Cas9 technology has been extensively used to create plants resistant to both abiotic stresses (adverse environmental conditions affecting plant growth) and biotic ones (pathogenic microorganisms). Recently, a group from the National Centre for Biotechnology in Madrid reported that the edition of the tomato SlJAZ2 gene related to the perception of the plant hormone jasmonate, helps protect these plants from pathogenic fungi via a mechanism associated with stomatal closure (Ortigosa et al., 2019). In cereals like rice, many of the improvements introduced using CRISPR/Cas9 have focused on developing plants that are more resistant to phytopathogenic fungi as well as improving their resistance to the high temperatures (Biswal et al., 2019) associated with climate change.
«On-demand genetic editing proves the enormous potential of these new strategies for the future improvement of crops»
An example of genetic editing which could have a major impact on human health is the development of wheat plants with a low content of gluten proteins responsible for several digestive disorders in humans (Sánchez-León et al., 2018). In this study, the authors designed two sgRNAs to edit conserved regions of the genes that encode alpha-gliadins, the main proteins that trigger sensitivity in individuals with coeliac disease. The approach was successful in introducing specific simultaneous mutations in this protein family, and they obtained plants with low gluten content and an 85 % decrease in immunoreactivity (Sánchez-León et al., 2018). This represents an unprecedented advance in obtaining commercial wheat varieties with low gluten content in the near future.
In recent years, our laboratory at the Institute for Plant Molecular and Cellular Biology (IBMCP, Valencia, Spain) has studied the process of fruit formation in tomato plants. Our studies revealed a mechanism by which the growth of non-pollinated ovaries is negatively regulated during the development of anthers. Consequently, the early removal of stamens results in the development of seedless fruits in this species (Medina et al., 2013; Rojas-Gracia et al., 2017).
Based on these studies, we used CRISPR/Cas9 technology to prove that the specific inactivation of genes responsible for the formation of pollen is sufficient to trigger the development of seedless (parthenocarpic) tomato fruits. Specifically, the insertion of a single nucleotide into a target gene sequence inactivates its function and results in pollen-free, parthenocarpic plants (Figure 3; unpublished results). This development could have applications in obtaining pollen-free ornamental plants as a containment method to help prevent their undesired spread or to reduce the presence of allergens. These new varieties are an example of on-demand genetic editing and prove the enormous potential of these new strategies for the future improvement of crops.
Notes
1. http://www.theplantlist.org/ (Go back)
2. CRISPR-Cas (CRISPR-associated) stands for clustered regularly interspaced short palindromic repeats. (Go back)
3. Protospacer adjacent motif. In the case of S. pyogenes, it has been determined that the PAM sequence is NGG, where N stands for any nucleotide and G stands for guanine nucleotide. (Go back)
References
Beltrán, J. P. (2018). Cultivos transgénicos. CSIC-Los libros de la Catarata.
Biswal, A. K., Mangrauthia, S. K., Reddy, M. R., & Yugandhar, P. (2019). CRISPR mediated genome engineering to develop climate smart rice: Challenges and opportunities. Seminars in Cell & Developmental Biology, 96, 100–106. http://doi.org/10.1016/j.semcdb.2019.04.005
Cong, L., Ran, F. A., Cox, D., Lin, S., Barretto, R., Habib, N., Hsu, P. D., Wu, X., Jiang, W., Marraffini, L. A., & Zhang, F. (2013). Multiplex genome engineering using CRISPR/Cas systems. Science, 339(6121), 819–823. http://doi.org/10.1126/science.1231143
FAO. (1983). World food security: A reappraisal of the concepts and approaches. Food and Agriculture Organization of the United Nations.
FAO. (1999). Women: Users, preservers and managers of agrobiodiversity. Food and Agriculture Organization of the United Nations.
FAO, IFAD, & WFP. (2012). The state of food insecurity in the world. Food and Agriculture Organization of the United Nations.
Gasiunas, G., Barrangou, R., Horvath, P., & Siksnys, V. (2012). Cas9-crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proceedings of the National Academy of Science of the USA, 109(39), E2579–2586. http://doi.org/10.1073/pnas.1208507109
Jinek, M., Chylinski, K., Fonfara, I., Hauer, M., Doudna, J. A., & Charpentier, E. (2012). A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science, 337(6096), 816–821. http://doi.org/10.1126/science.1225829
Lander, E. S. (2016). The Heroes of CRISPR. Cell, 164(1-2), 18–28. http://doi.org/10.1016/j.cell.2015.12.041
Liang, Z., Chen, K., Li, T., Zhang, Y., Wang, Y., Zhao, Q., Liu, J., Zhang, H., Liu, C., Ran, Y., & Gao, C. (2017). Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9 ribonucleoprotein complexes. Nature Communications, 8, 14261. http://doi.org/10.1038/ncomms14261
Medina, M., Roque, E., Pineda, B., Cañas, L., Rodríguez-Concepción, M., Beltrán, J. P., & Gómez-Mena, C. (2013). Early anther ablation triggers parthenocarpic fruit development in tomato. Plant Biotechnology Journal, 11(6), 770–779. http://doi.org/10.1111/pbi.12069
Metje-Sprink, J., Menz, J., Modrzejewski, D., & Sprink, T. (2018). DNA-free genome editing: Past, present and future. Frontiers in Plant Science, 9, 1957. http://doi.org/10.3389/fpls.2018.01957
Mojica, F. J., Díez-Villaseñor, C., García-Martínez, J., & Almendros, C. (2009). Short motif sequences determine the targets of the prokaryotic CRISPR defence system. Microbiology, 155(Pt 3), 733–740. http://doi.org/10.1099/mic.0.023960-0
Mojica, F.J, & Montoliu, L. (2016). On the origin of CRISPR-Cas technology: From prokaryotes to mammals. Trends in Microbiology, 24(10), 811–820. http://doi.org/10.1016/j.tim.2016.06.005
Montoliu, L. (2019). Editando genes: recorta, pega y colorea. Las maravillosas herramientas CRISPR. Next Door Publishers.
National Academies of Sciences & Medicine. (2016). Genetically engineered crops: Experiences and prospects. The National Academies Press.
Ortigosa, A., Giménez-Ibáñez, S., Leonhardt, N., & Solano, R. (2019). Design of a bacterial speck resistant tomato by CRISPR/Cas9-mediated editing of SlJAZ2. Plant Biotechnology Journal, 17(3), 665–673. http://doi.org/10.1111/pbi.13006
Osakabe, Y., Liang, Z., Ren, C., Nishitani, C., Osakabe, K., Wada, M., Komori, S., Malnoy, M., Velasco, R., Poli, M., Jung, M.-H., Koo, O.-J., Viola, R., & Nagamangala Kanchiswamy, C. (2018). CRISPR-Cas9-mediated genome editing in apple and grapevine. Nature Protocols, 13(12), 2844–2863. http://doi.org/10.1038/s41596-018-0067-9
Rojas-Gracia, P., Roque, E., Medina, M., Rochina, M., Hamza, R., Angarita-Díaz, M. P., Moreno, V., Pérez-Martín, F., Lozano, R., Cañas, L., Beltrán, J. P., & Gómez-Mena, C. (2017). The parthenocarpic hydra mutant reveals a new function for a SPOROCYTELESS-like gene in the control of fruit set in tomato. New Phytologist, 214(3), 1198–1212. http://doi.org/10.1111/nph.14433
Sánchez-León, S., Gil-Humanes, J., Ozuna, C. V., Giménez, M. J., Sousa, C., Voytas, D. F., & Barro, F. (2018). Low-gluten, nontransgenic wheat engineered with CRISPR/Cas9. Plant Biotechnology Journal, 16(4), 902–910. http://doi.org/10.1111/pbi.12837
Shan, Q., Wang, Y., Li, J., Zhang, Y., Chen, K., Liang, Z., Zhang, K., Liu, J., Xi, J. J., Qiu, J.-L., & Gao, C. (2013). Targeted genome modification of crop plants using a CRISPR-Cas system. Nature Biotechnology, 31(8), 686–688. http://doi.org/10.1038/nbt.2650
Taylor, S. L., & Hefle, S. L. (2001). Ingredient and labeling issues associated with allergenic foods. Allergy: European Journal of Allergy and Clinical Immunology, 56(67), 64–69. http://doi.org/10.1034/j.1398-9995.2001.00920.x
Wang, T., Zhang, H., & Zhu, H. (2019). CRISPR technology is revolutionizing the improvement of tomato and other fruit crops. Horticulture Research, 6(1), 77. http://doi.org/10.1038/s41438-019-0159-x
Wolter, F., Schindele, P., & Puchta, H. (2019). Plant breeding at the speed of light: the power of CRISPR/Cas to generate directed genetic diversity at multiple sites. BMC Plant Biology, 19(1), 176. http://doi.org/10.1186/s12870-019-1775-1





