 The future of food lies in personalized nutrition, that is, smart diets designed according to the specific demands of individual genotypes and their history / © M. Lorenzo |
||
|
Nutrigenomics: personalized nutrition. Knowledge about the relationship between health and food is currently increasing. The relationship between our welfare and longevity with the biochemical diversity of the food we eat, as well as other environmental conditions, is widely known. At the same time, people are more aware of the fact that the wide differences that exist between individuals in the response to food depend on the particular traits encoded in our genes and on the adaptations implemented in the different conditions experienced by each person throughout their lives. This gave rise to nutrigenomics, an emerging discipline that studies how food and its components interact with the information encoded in our genes and with the entire metabolic network derived from it, as well as their consequences at all levels. Nutrigenomics seeks extensive and integrated knowledge of how diets or their components affect biological systems at all levels, from the expression of genetic information that characterizes them (transcriptome), their protein composition (proteome) to the different biomolecules or metabolites that constitute them (metabolome) and how all that translates into the operation of these systems and affects the whole organism. Nutrigenomics is also responsible for understanding and characterizing the different response to foods depending on the genotype (nutrigenetics) and on the individual dietary history (epigenetics). Nutrigenetics and nutrigenomics are often considered two different disciplines. Nutrigenetics, or traditional nutrigenomics, seeks to understand how genetic characteristics of an individual determine their response to a diet, and to achieve this, this discipline is based on the consideration of the underlying polymorphisms. By encompassing a wider field, nutrigenomics tries to determine the influence of food on the genome, and tries to relate the resulting phenotypes with the different genetic, cellular or physiological response and, in general, with the response of the biological system. Therefore, it also includes the study of the differential response associated with particular genotypes (nutrigenomics) and phenotypes. One of the ultimate goals of nutrigenomics is to develop foods that meet the health conveniences of individuals. Actually, the real underlying challenge is integrating all information, that derived from the data and knowledge provided by the new omics technologies (genomics and post-genomics), with that provided by classical nutrition and human metabolism; everything that can reveal the effects of an external agent on an organism. No wonder, then, that new terms and concepts such as «systems biology», «integrated metabolism» or «nutritional systems biology» arise as progress is made, trying to define specific fields of knowledge. Here we consider that the term «nutrigenomics» is quite broad and appropriate to encompass, at least for now, all these new developments in the field of nutrition and health to which we have referred. Actually, knowledge about nutrigenomics has been on the scene for many years and its applications are widespread; such as when we restrict the diet of people affected by certain diseases due to single gene mutations or variants (so-called inborn errors of metabolism) or we prescribe not eating certain foods to certain population groups who, unlike others, present adverse reactions against a particular food component. However, the framework that configures nutrigenomics is much broader: it takes into account polymorphisms (different gene variants), in which the presence of a specific allele means only some difference, however small, in relation to particular function, and also the combinations of multiple variants that, individually treated, may not have any functional effect but which in combination do determine new properties (Palou et al., 2004). Moreover, it includes the metabolic history of each person, the result of exposure to various nutrients and other factors during the different stages of life (epigenetics); certainly this individual history also contributes significantly to a likewise individual response. Finally, it takes into account the mechanisms involved in the effects of nutrition and their significance at all phenotypic levels and, in particular, how they affect the scale that, on both sides, leads to function or dysfunction, health or disease. |
«Nutrigenomics seeks extensive and integrated knowledge of how diets or their components affect biological systems at all levels» | |
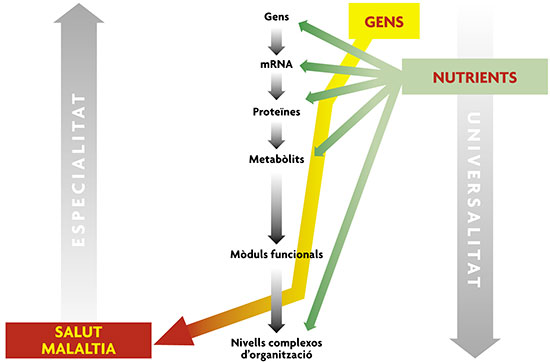 © Mètode The effects of food on biological systems can be described by nutrigenomics information (genetic profile, expression of messenger RNA, proteins and metabolites). These effects may include changes or shifts in the system that controls body weight and hence contribute to obesity. Nutrigenomics seeks, based on the description of the genes / gene expression / proteins / metabolites in a person, to predict the possible homeostatic deviations in healthy people, that is, before the systems had been irreversibly biased towards the development of illnesses in the short, medium or long term. (Adapted from Oltvai and Barabasi, 2002.) |
«Nutrigenomics is responsible for understanding and characterizing the different response to foods depending on the genotype (nutrigenetics) and on the individual dietary history (epigenetics)»
«Actually, knowledge about nutrigenomics has been on the scene for many years and its applications are widespread» |
|
| NEW TECHNOLOGIES IN NUTRIGENOMICS
Nutrigenomics uses traditional techniques in metabolism and nutrition but also new biochemical technologies and, in particular, the so-called omics technologies (transcriptomics, proteomics, metabolomics). It is nourished by the rapid advances in our knowledge of the genes composing the genome, their regulating mechanisms and how certain food components influence these systems; and benefits from the great advances being made in human biochemistry and particularly metabolism. This way, nutrigenomics provides an added value to epidemiological knowledge and the classic contents of nutrition, giving this science a solid predictive capacity. These modern technologies have capitalized on the wealth of information provided especially by the sequencing of the human genome; the study of genes, proteins and metabolites and their relationship with other components and cellular and tissue parameters have resulted in the adoption of many denominations (over 50) of techniques or technologies ending with the suffix -omics or -ome (Greek for “totality”) appearing in scientific literature. The SNP (Single Nucleotide Polymorphisms) consortium is locating numerous polymorphisms in the human genome that determine individual differences in phenotypic traits found in the population. Genomics can be described as the study and characterization of the sequence of all genes in a given species. Several areas derive from it, such as «functional genomics», which covers aspects of knowledge of the functions of the different genes and how these genes interact and influence each other, and transcriptomics, which uses cDNA or oligonucleotides microchip technology (microarrays) as approaches to analyse gene expression (thousands of mRNA can be identified in any biological sample under certain conditions). The transcriptome is the set of all the mRNA from a fluid, cell, tissue or biological sample in general. Right now it is the most abundantly used omics technology and the one with better chance of validation and standardization for virtually all mRNA transcripts, although a relatively large sample is needed to isolate a fair amount of the mRNA that is needed. Proteomics aims to identify all the proteins in a biological sample, including the relative abundance, distribution, post-translational modifications, functions and interactions with other biological molecules and, although results are being obtained, the technological developments that are currently taking shape will change it into a very powerful technique. Metabolomics (or metabonomics, less preferable here because it is more associated to the profile of metabolites in drug studies) can be defined as the quantitative analysis of all the metabolites, endogenous and exogenous, of a particular biological fluid, tissue, or, in general, of a particular biological system. Regardless of whether proteomics and, especially, metabolomics require further studies that provide us with standard and routine procedures widely applicable for the complete development of the goals of these techniques in nutrition, we need more mathematical tools. At present, we do not have the appropriate treatments for the many thousands of data generated by studies and, while it is difficult to specify, we believe that less than 2% of the information obtained is processed; the rest is lost. This makes it necessary to implement strategies for selecting the parameters to be analyzed, which allows to reduce the cost of processes, although it is not clear that this is the most appropriate option. A pending fundamental tool is the establishment of omics profiles defining «standard health» states that can serve as a reference or control in different studies. Similarly we can refer to the establishment of nutrigenomic profiles for other conditions such as diabetes mellitus type 2, obesities and other diseases or physiological conditions of interest (see below). One of the biggest difficulties is found when analyzing data sets and variables combined, taking also into account that they correspond to different coordinates of a multidimensional space defined by different fields (genetic, clinical, nutritional, metabolic, metabolomic, etc., and other variables) that reflect, each with specific laws and guidelines, the information on homeostatic processes and the balance between health and disease. Determining the useful nutritional health phenotypes (see below) is one of the biggest challenges. NUTRIGENOMIC BIOMARKERS A large number of reviews already illustrate the ability to identify at an early age sensitivities of certain people to diet-related diseases, which could strengthen (along with dietary and lifestyle advice, and knowledge of diet-genotype interactions) the benefits of diet on human health and advance knowledge of the scientific basis of the relationship between diet and health. Nutrigenomics tries to describe the biological system (the operation of a cell, tissue, biological system in general, or our entire organism in the broader case at hand) in «its totality»; in terms of their composition of thousands of mRNAs, proteins and metabolites, and the combinations of all these. With that we would like to «completely» describe each homeostatic system to perceive shifts in it, prior to the appearance of abnormalities or risk factors that may eventually lead to some more definite abnormalities (diseases or other phenotypes that interest us). In this way, nutrigenomic biomarkers result from sets of characteristic data of the response (to a particular diet or food composition or dietary pattern) of the biological system, indicative of the system’s derivation to a certain considered potentially adverse condition; for example, to the development of hypertension. There are already numerous nutrigenomics studies showing the relationship between dietary components and health. Examples include the role of fatty acids in the development of hepatic steatosis or fatty liver, the effect of calcium and hemo on the risk of colon cancer, or the effects of olive oil, alcohol, vitamin D, etc. It is logical that a complete profile of many biomarkers, based on hundreds or thousands of parameters, more faithfully represents the state of health or welfare than the current and few biomarkers routinely available. Among the current difficulties to be overcome is the fact that, at present, a relatively large amount of sample is required to isolate enough material for analysis and the fact that, in general, access to human tissues (apart from blood, urine and other fluids and peeling, easily practicable) is limited. Although biopsies of skin, muscle and adipose tissue are relatively accessible and simple, the different types of blood cells offer a priori the most interesting possibilities. In the future we expect the development of new techniques for noninvasive analysis. It is possible to determine characteristic tissue profiles and identify candidate genes, for example in relation to obesity, or markers of the effects of nutrients, such as vitamin D, or identify several agents and contaminants of interest in food safety. Specific patterns of gene expression in blood cells associated with diseases such as lung tumors or leukemia that can serve as markers of disease have been described, but the question on pre-disease biomarkers, before the onset of the factors and risk signs traditionally considered, is still pending. |
||
 © M. Lorenzo Exposure to tobacco or physical exercise may be considered as minor factors, though still important, when compared with the continued exposure to food throughout our lives. |
«One of the ultimate goals of nutrigenomics is to develop foods that meet the health conveniences individuals» | |
|
So far, although many experiences have not been published and therefore have not been shared (a problem that the scientific community should solve, particularly publishers), we can deduce that the interindividual variability observed is high, while it is true that with the data obtained it is feasible to establish concordance; for example, we are already obtaining quite reproducible resuts by adjusting the conditions, daily and seasonal rhythms, forms of nutrition, etc. of each individual. In this way, nutrigenomic profiles are presented as a promising strategy to advance the understanding of phenotypic response to nutritional interventions. ON RECOMMENDATIONS IN CURRENT NUTRITION In general, it is worth noting that exposure to tobacco or other toxins, or physical exercise, which we know are very capable agents determine our health, are factors that can be considered minor when compared to our continued exposure to food throughout our lives. Food is the main environmental factor that tests our body. The benchmarks of quality in food have changed and consumer demands change very fast. Concerns during the past century first focused on ensuring the availability of basic foods and, later, on making sure they were safe. In nutrition, they were focused on preventing nutritional deficiency diseases, ensuring an adequate supply of macronutrients and essential components. Today, however, our society is more focused on addressing (and in a key way through food) the so-called chronic diseases of our time: heart disease, diabetes, obesity, various cancers, osteoporosis and autoimmune diseases. The consumer in our developed societies, with an increasing purchasing power, does not only demand food to be safe, but also available, and will not be satisfied with the traditional expectations of food (energy, nutrients for the construction or renovation of our body structures, that they satisfy our hunger and the pleasure of eating, their role in social relationships and traditions, that their production respects animal welfare and the environment). Besides all that, they expect food to provide them more health and welfare (functional foods). European food law adapts to new scenarios. The extensive debate stirred up by the proposal on Nutrition and Health Claims Made on Foods (see Regulation (EC) no. 1924/2006 on nutrition and health claims made on foods, Official Journal of the European Union L 12, 3-18, 18 January, 2007), ended in Europe in late 2006. This was an initiative of the EC on the regulations that should govern uniformly all member states to reduce the current situation of anarchy in health-food advertising (each country had its own interpretation – often oscillating – about the properties or benefits that may or may not be alleged in tags, ads, etc.), which promotes the proliferation of misleading advertising, not supported by scientific evidence, and affects all consumers, including children and young people. The new legislation bans nutritional or health claims for foods that do not have a minimally healthy «nutritional profile» (foods high in saturated fat, salt, etc.), profile that should be evaluated independently by a panel of nutrition of the EFSA (European Food Safety Authority). The underlying idea is that only scientifically tested health claims must be allowed, and since health claims may stimulate the consumption of those foods that have them, it doesn’t seem reasonable that claims benefit the consumption of foods with an inappropriate nutrient profile. Maybe it is, for now, more politically correct to keep saying there are no good or bad foods (because it is true that the effects depend on the dose); but scientific knowledge brings each day further evidence that there are marked differences between the different foods, which should not be ignored. In addition, although dietary recommendations have already been introduced today to improve health and reduce the risk of CVD, cancer, hypertension, type 2 diabetes, obesity, and other chronic diseases, with tested benefits according to statistics, it is true that these recommendations have been established on the basis of the general population and not individually. Without disregarding the benefits in general, such drastic differences in the responses of each individual to a particular diet show the limitations of these general recommendations, and also our very partial knowledge about these mechanisms. Parallel to the consumer’s better nutritional training and information, the changes will give way from a «paternalistic» position of administrations (with general recommendations for a healthy and varied diet in general that ensure – with food reinforcement, if necessary – the absence of dietary deficiencies) to a situation in which the consumer makes an informed decision of the foods and ingredients of the diet that better fit their health and welfare. FUNCTIONAL FOODS AND SMART DIETS The capitalization of knowledge about new health and welfare benefits being shaped is the basis for the development of so-called functional foods. Certain food components and diets are able to decide our body’s adjustments as they affect the maintenance of homeostatic equilibriums that determine health and welfare conditions in the sense of favouring or preventing certain chronic diseases or other conditions of interest. |
||
 © M. Lorenzo Modern consumers are not satisfied with the availability and safety of food, they also want food to provide them with health and welfare. |
«Nutrigenomics will improve both the safety and efficiency of food» | |
|
Nutrigenomics will improve both the safety and efficacy of foods because it will provide a more precise level of understanding of the influences of foods and food components in our homeostatic systems, with new approaches to the determination of effects, beneficial or adverse, in early stages; for example, before the development of a disease. That includes diets aimed at population subgroups and even personalized nutrition, smart diets designed according to the specific demands of individual genotypes and their history. For example, we can think that (and there is already reckless commercial propaganda about it) that from a small sample of blood or from other readily available sample we can perform (and easily, quickly and at an acceptable cost) a nutrigenomics survey (transcriptomics, proteomics, metabolomics), compare the results with the DNA of the individual (also measurable) and thereby obtain an intelligent selection of varied, healthy diet (recommended, for example, for the next twenty days), balanced in macronutrients and micronutrients and with the optimal quantity of bioactive compounds, plus tasty. The main difficulties to overcome are derived from the complexity of food and eating practices themselves, and also from the complexity of our metabolic systems and their subtle inter-regulations. We must also take into account the problems of social perception, ethical issues and economic and social implications. We are currently gathering a lot of information; genotyping of individuals participating in dietary intervention studies has been incorporated into many of the large-scale study protocols; large-scale observational studies on the relationships between diet and disease also are including the research on the interactions between diet and genotype. The concept of individualized nutrition, dietary advice tailored to each individual, has captured the imagination of nutrition researchers in both public and therapeutic health. But we have said imagination, not immediate applicability and commercialization. The opportunities for the food industry in providing specific functional foods and products tailored to subgroups according to specific genotypes have also been recognized, and the general dogma that says that a diet «fits all foods» has begun to be cast into doubt. The opportunities of personalized nutrition, as well as the ethical and social implications derived from it, have attracted the attention of the general media. Considering current trends and the availability of all types of ready-made foods that accurately reflect the taste and smell of their original counterparts, we should face the knowledge of the nutrigenomics response to these complex preparations, and not only the knowledge about the nutrigenomic profile of individual food nutrients or components. |
||
|
EPIGENETICS AND DEVELOPMENT OF DISEASES Food, particularly during the early stages of development, can strongly determine our health throughout life. Thus, the prospective mother’s diet is a primary factor – more than previously thought – in the development of children, especially in regard to their future health and disease resistance. We know today that some diseases that occur in adulthood, and that were attributed to an unhealthy lifestyle or genetic factors, are a direct result of the uterine environment during pregnancy or during lactation. Several large epidemiological studies conducted in the last twenty years have shown that nutritional conditions during critical stages of development, especially gestation and early postnatal period (mainly lactation) affect the susceptibility of developing certain diseases in adulthood, including cardiovascular disease, obesity, type 2 diabetes, osteoporosis and other problems and disorders. The underlying biological mechanisms are not well known, however, as is clear from studies in animal models or in vitro, the differences are caused by the interaction between food components (or other environmental factors) and our chromosomes, which lead to imprinting or the programming of metabolic patterns particular to individuals, including permanent changes (epigenetic) transmitted in cell division and that may confer different susceptibility to suffering disorders in adulthood. Studies conducted in recent years in our laboratory have allowed us to describe a new role for one of these proteins found naturally in breast milk and not present in infant formulas: leptin, whose new role during breastfeeding is to confer protection against the development of overweight or obesity (and other associated medical complications) in adulthood. The term imprinting, or metabolic programming, describes the processes by which cells acquire a certain biological memory to handle external influences, and which can be transmitted to cell progeny. It implies that a stimulus that operates at a critical or sensitive period of development (which could be considered as a specific window of sensitivity) can lead to persistent or permanent effects on the structure or function of the body. Epigenetic regards these stable alterations in gene expression that do not involve DNA mutations in itself but which confer a particular pattern of gene expression that remains stable during cell divisions. Methylation has been considered to be the main epigenetic basis for metabolic programming; histones, proteins closely associated to DNA, may contribute to these epigenetic changes and DNA methylation would be related to histone modification. Although leptin is mainly produced by adipose tissue, this hormone is also produced by other tissues such as the placenta and skeletal muscle and, in particular, by the stomach and the breast epithelium, and it is naturally present in breast milk. Leptin concentrations in breast milk vary significantly between mothers, but it is significant that artificial milks or infant formulas do not contain leptin as an ingredient. Breast milk is virtually the only food eaten during the first months of life by infants fed exclusively with breast milk and, compared with formula, the results of several epidemiological studies indicated that breastfeeding confers greater protection against obesity and other disorders in adulthood. Studies have also shown an inverse correlation between the duration of breastfeeding and risk of overweight in adulthood. Leptin receptors are present in the human stomach and in the gastrointestinal tract of mice, and leptin is able to directly activate vagal afferent neurons that originate in the gastric and intestinal walls and end up in the nucleus of the solitary tract, providing quick information to the brain. Furthermore, it was reported that leptin supplied with milk, or administered orally in aqueous solution, could be absorbed by the immature stomach of lactating rats and pass into blood circulation. In addition, leptin supplied with human breast milk appeared as the main source of leptin in the stomach during the first half of infancy. This exogenous leptin could exert biological effects in newborns, in a period in which both the adipose tissue and the hunger-regulating systems are immature. Thus, we will consider the hypothesis that leptin could be the specific compound (or at least one of them) responsible for the beneficial effects of breast milk in the protection against obesity at older ages. To prove it, we supplemented lactation in rats with physiological doses of leptin administered orally and observed the possible effects that occurred on the regulation of body weight in adulthood. The results showed that the animals that had taken leptin during lactation had less weight gain and less fat accumulation in adulthood, and ate fewer calories than their untreated counterparts, both under standard diet conditions and in high-fat diets. The overall conclusion of the study was that supplementation with physiological amounts of leptin during lactation protects against the accumulation of fat in adulthood and that these animals seem more sensitive to the regulation of food intake by leptin, in the short and long term. Thus, leptin plays an important role in the early stages of life, as a component of breast milk, in the prevention of obesity later in life. Other studies carried out on humans have confirmed these and other findings on the susceptibility to other diseases related to obesity. Moreover, the results obtained in non-obese mothers and their children up to two years of age indicate that larger intake of leptin in breast milk provides some protection for children against excessive weight gain. Sequencing the human genome has revealed not only a lower number of human genes than expected (the latest estimations show about 24,000 genes), but also the great genetic heterogeneity of human populations, a greater genetic variability among humans than previously assumed: it is estimated that the human genome contains about ten million polymorphisms. This complements the complexity arising from the extensive interactions between genes and their regulation, as well as epigenetic variability, the «metabolic imprinting»: our story of how we eat, how we feel, environmental influences, etc., is recorded permanently. All this promotes a deep change in human thought on how this information can be exploited optimally for the benefit of health and well-being. The role of genetic polymorphisms in determining heterogeneous responses to environmental exposures such as medications, physical activity and diet, is already evident. The pharmaceutical industry is already exploring this opportunity through the development of pharmacogenomics and, in the same way, the food industry will have to be involved in the development of nutrigenomics. The opportunity to develop nutrigenetic or nutrigenomics tests commercially seem real, in fact there is a growing segment of consumers who demand them and who is able to bear the costs, despite the existing doubts; that’s why the tests to which consumers can now go directly are increasingly accessible, although we believe that this commercialization is still premature. |
 © M. Lorenzo The interaction of food components with our chromosomes during the stage of pregnancy and the first months of life are extremely important to our future life, since it will condition the diseases we will suffer in adulthood. «We know today that some diseases that occur in adulthood, and that were attributed to an unhealthy lifestyle or genetic factors, are a direct result of the uterine environment during pregnancy or during lactation» |
|
REFERENCES
Casabiell, X. et al., 1997. «Presence of leptin in colostrum and/or breast milk from lactating mothers: a potential role in the regulation of neonatal food intake». J. Clin. Endocrinol. Metab., 82: 4270-4273.
Darnton-Hill, I. et al., 2004. «Public health nutrition and genetics: implications for nutrition policy and promotion». Proc. Nutr. Soc., 63: 173-185.
Elliott, R. et al., 2007. «Nutrigenomic approaches for benefit-risk analysis of foods and food components: defining markers of health». Br. J. Nutr, 1-6.
Harder, T., 2005. «Duration of breastfeeding and risk of overweight: a meta-analysis». Am. J. Epidemiol., 162: 397-403.
Mutch, D. M. et al., 2005. «Nutrigenomics and nutrigenetics: the emerging faces of nutrition». Faseb J., 19: 1602-1616.
Oltvai, Z. N. and A. L. Barabasi, 2002. «Systems biology. Life’s complexity pyramid». Science, 298: 763-764.
Palou, A. et al., 2004. «Nutrigenomics and obesity». Rev. Med. Univ. Navarra, 48: 36-48.
Sánchez J. et al., 2005. «Leptin orally supplied to neonate rats is directly uptaken by the immature stomach and may regulate short-term feeding». Endocrinology, 146: 2575-2582.





