Evolution of the ‘Homo’ genus
New mysteries and perspectives

This work reviews the main questions surrounding the evolution of the genus Homo, such as its origin, the problem of variability in Homo erectus and the impact of palaeogenomics. A consensus has not yet been reached regarding which Australopithecus candidate gave rise to the first representatives assignable to Homo and this discussion even affects the recognition of the H. habilis and H. rudolfensis species. Regarding the variability of the first palaeodemes assigned to Homo, the discovery of the Dmanisi site in Georgia called into question some of the criteria used until now to distinguish between species like H. erectus or H. ergaster. Finally, the emergence of palaeogenomics has provided evidence that the flow of genetic material between old hominin populations was wider than expected.
Keywords: palaeogenomics, Homo genus, hominins, variability, Dmanisi.
In recent years, our concept of the origin and evolution of our genus has been shaken by different findings that, far from responding to the problems that arose at the end of the twentieth century, have reopened debates and forced us to reconsider models that had been considered valid for decades. Some of these questions remain open because the fossils that could give us the answer are still missing. Conversely, others are still the subject of debate because of the different interpretations and systematic allocations given to some fossils. Findings such as those from the Dmanisi site in Georgia or those provided by palaeogenomics, especially put into question the criteria used so far to recognise species in the fossil record; some of these issues are discussed below.
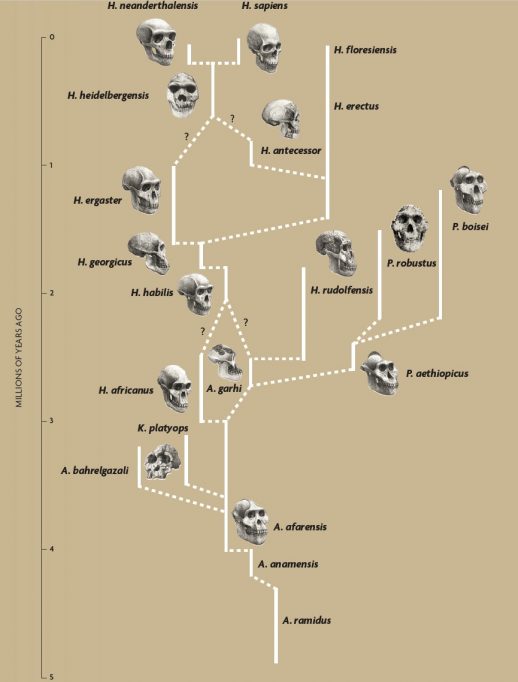
A diagram of human evolution, as proposed by Agustí and Lordkipanidze (2005). In recent years, our conception of the origin and evolution of the Homo genus has been shaken by different findings that have reopened debates and forced us to rethink certain paradigms. / Skull illustrations: Mauricio Antón
The origin of the ‘Homo’ genus
The first representatives of our genus appeared in the African fossil record about 2.8 million years ago (Villmoare et al., 2015) and these first remains have sometimes been attributed to Homo rudolfensis. We also have evidence of a second species, Homo habilis, which lived a little more than two million years ago; this species differs from H. rudolfensis in some secondary characteristics and in its smaller cranial capacity, although some researchers believe that Homo habilis and Homo rudolfensis correspond to the same species.
Until the mid-1970s, there was a clear Australopithecine candidate to occupy the position of our genus’ ancestor, Australopithecus africanus, which is exclusively distributed in the south of Africa. However, the 1978 publication of data from the remains of an Australopithecus afarensis specimen from Afar in Ethiopia, put this connection into question for the first time. Its discoverers, Don Johanson and Tim White, posited that Australopithecus afarensis dates to between 3.5 and 3 million years and fits in perfectly with our genus, while Australopithecus africanus would have given rise to the robust forms of Australopithecines found in the south of Africa and framed within the Paranthropus genus.
Later, in 1996, White proposed a new species, Australopithecus garhi, found at levels dating back some 2.5 million years in Bouri, Ethiopia. According to White and his collaborators, A. garhi was in the right place at the right time to be the ancestor of our genus. Bones associated with A. garhi were also found and these showed cut marks from lithic tools which were thought to have been produced by this species.
To complicate matters, in 2001 the team led by Meave Leakey proposed another new genus, Kenyanthropus, for a very deformed skull from the Lomekwi Basin in Kenya that dates back some 3.5 million years (i.e., contemporary to A. afarensis). The estimated cranial capacity of the so-called Kenyanthropus platyops was low, at around 350 cm3. But what led its discoverers to propose a new genus was its face, which was long and flat. Taking a further step, they proposed that this form was the ancestor of Homo rudolfensis, a species that they proposed should also be included in the Kenyanthropus genus. This latter proposition has found little response among the paleoanthropological community, which, far from accepting the new genus, chose to include the species K. platyops within Homo or Australopithecus. However, some authors have accepted the suggestion of a relationship between K. platyops and H. rudolfensis.
Beyond the findings in east Africa, the idea of a south African origin for our genus came back to the fore with the discovery of a new species of Australopithecus, A. sediba, in the Malapa cave in South Africa, where two partial skeletons belonging to a young woman in her twenties and an adolescent around twelve years old were found. The most significant element is a skull in an excellent state of conservation, belonging to an Australopithecus close to Australopithecus africanus but that has more derived characteristics. Apart from the fact that it had a somewhat larger brain than its predecessors’, the face shows some features much closer to the first representatives of our genus (such as Homo habilis), for instance, the shape of the nasal cavity and cheekbones. For its discoverer, Lee Berger, from the University of Witwatersrand (South Africa), Australopithecus sediba is the perfect intermediate link between Australopithecus africanus and the first representatives of the genus Homo. The problem, however, is that the Australopithecus sediba remains date back about 1.8 million years and therefore correspond to way after the emergence of our genus over 2.5 million years ago. The same team recognised a new archaic species of the genus Homo, H. naledi, in the Dinaledi Chamber, a very difficult-to-access cavity also located in South Africa. However, the absence of an adequate geological and palaeontological context calls into question the possible relevance of this finding in relation to the origin of our genus.

Homo rudolfensis was considered the first representative of the genus Homo. Its remains date back to 2.8 million years ago. We might have evidence of the existence of a second species, H. habilis, from a little more than two million years ago, although some researchers consider both H. rudolfensis and H. habilis to be the same species. The image shows a skull and a reconstruction of H. rudolfensis (according to Mauricio Antón). / Illustration: Mauricio Antón
Regarding the Australopithecine candidates in east Africa, some surprising data emerged: they tend to have large molars (a phenomenon known as «megadontia»), which would later peak in the different species included in the Paranthropus genus. This trend is already evident in Australopithecus afarensis, a species that may be a good fit as an ancestor of Homo, but also as a plausible ancestor of Paranthropus. In fact, when the so-called «black skull» of the Paranthropus aethiopicus came to light, several authors noted its similarities to Australopithecus afarensis. If we now look at Australopithecus garhi, this tendency towards megadontia is even clearer, although cranial morphology does not allow us to include it in the Paranthropus genus. However, it is evident that east Africa offered an environmental context favouring the development of a powerful masticatory apparatus and large molars.
Palaeontologists subsequently went on to analyse the dentition of the first members of our genus, and reached the surprising conclusion that the first remains assigned to Homo also had large molars, which is why they are commonly included within the species Homo rudolfensis. The remains assigned to Homo habilis, whose dentition was more graceful and closer to that of the south African Australopithecus, appeared later.
This is therefore a dichotomy; in east Africa we could recognize a lineage leading to Homo rudolfensis, through Australopithecus afarensis and Australopithecus ghari (and perhaps Kenyanthropus), while on the other hand, there would be an elegant line leading to Homo habilis from Australopithecus africanus, and perhaps including Australopithecus sediba. Thus, the surprising conclusion is the potential diphyletic origin of the Homo genus.
However, given the fragmented nature of the register corresponding to the first Homo genus variants, we might wonder whether both Homo habilis and Homo rudolfensis are in fact variants of the same species. The differential elements between the two may be a consequence of the population variability within that species.
The status of ‘Homo erectus’ and the question of variability
In the mid-1970s, two conceptual revolutions in the field of evolutionary biology made a profound mark on ideas about our own evolution. On the one hand, in the palaeontological system, the use of cladistics for establishing the phylogeny of any fossil group became widespread. In this way, with its distinction between primitive and derived characteristics, it was possible to develop verifiable hypotheses about the relationship between different taxa.

In 2001, the team of paleoanthropologist Meave Leakey proposed the new genus Kenyanthropus. For these researchers, Kenyanthropus platyops was the ancestor of Homo rudolfensis, a proposal that has not resonated much with the scientific community. The image shows a skull and a reconstruction of K. platyops (according to Mauricio Antón). / Illustration: Mauricio Antón
Even more influential was the proposal of the palaeontologists Niles Eldredge and Stephen J. Gould that a large part of evolutionary sequences does not fit the simplistic model of gradual change; they hypothesised that species appeared abruptly in moments of sudden change and remained stable until their extinction. In the field of human evolution, Niles Eldredge and Ian Tattersall picked up the gauntlet. In 1982, they published The myths of human evolution, a work that would later have a strong influence on our conception of evolutionary patterns in human evolution. From then on, the fact that human evolution had been a much more varied and complex process became evident in the interpretation of old fossils, as well as in any new ones. The landscape of human evolution suddenly changed and under the new paradigm new species came to the fore: Homo rudolfensis, Homo ergaster, Kenyanthropus platyops, Homo antecessor, Homo georgicus and others. Some previous species exploded, such as the classic Homo erectus, which ceased to be a necessary link in human evolution and was restricted to the Asian context. This paradigm remained in force during the 1980s and part of the 1990s. At the end of this decade, however, there were early signs of disagreement by researchers such as Tim White, who questioned the distinction between Homo erectus and Homo ergaster. But the findings in the Georgian site of Dmanisi are the ones that will once again bring about a change in the prevailing paradigm.
Over the last few decades, the Dmanisi site, dated by radiometric and palaeomagnetic methods back to 1.8 million years, has provided the best fossil record of the first hominids that left Africa (Agustí & Lordkipanidze, 2005). It all began in 1991, when the first piece of human remains, a jaw that retained the entire dentition, was found in Dmanisi. The jaw was debated from the beginning. Some authors questioned its age based on the degree of reduction of the posterior molars, and placed it in the middle, rather than the lower, Pleistocene. It was not until 1999 that the discovery of two new skulls confirmed the archaic character of the Dmanisi hominins. In the first, D2280, only the cranial calotte, i.e., the highest part of the skull that houses the brain, was conserved, but this allowed experts to infer a cranial capacity of 775 cm3. The second, D2282, was very deformed, had also retained part of the facial bone, and its cranial capacity was estimated at about 650 cm3. In the year 2000 a new jaw appeared, but this time a large one with highly-developed posterior molars, which corresponded to an archaic representative of the Homo genus. Other features of the jaw also evidenced clear primitivism, and so the Georgian palaeontologist Leo Gabunia and his team decided to form a new species, Homo georgicus. This jaw gave rise to the first speculations regarding the presence of two hominin species in Dmanisi.
The following year, in 2001, a new skull came to light in Dmanisi. This time, the D2700 skull included the jaw and was in an almost perfect state of conservation. The detailed analysis of this skull showed a very archaic hominin, with a cranial capacity of 600 cm3. The profile was concave and, unlike the first skull, D2280, this one had underdeveloped superciliary arches and the jaw resembled the first jaw from 1991 (Vekua et al., 2002). The D2700 skull clearly demonstrated that the Dmanisi hominins could not be assigned to the African species Homo ergaster and that the first departure from Africa was led by populations closer to Homo habilis than to the latter species. This attribution was confirmed in 2002, when a new senile skull appeared: a toothless individual (the corresponding jaw appeared the following year, in 2003), which nevertheless showed the same archaic features as the 2001 skull (Lordkipanidze et al., 2005). The 2005 campaign finally led to the spectacular discovery of the D4500 skull, which matched the large mandible from the year 2000. This skull did not betray the expectations created by the discovery of the corresponding jawbone. Although its cranial capacity was the lowest of the entire Dmanisi population, at only 546 cm3, its robustness surpassed previous findings (Lordkipanidze et al., 2013).
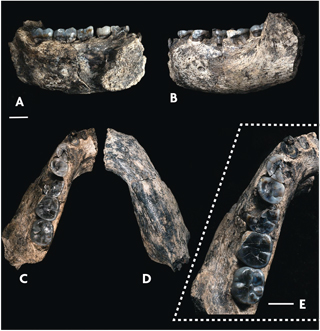
View of a fossil jawbone from different perspectives. / Villmoare et al., 2015
The analysis of the complete Dmanisi palaeodeme led the paleoanthropological team to conclude that, despite its variability, the set corresponded to a single population. The geological, palaeomagnetic, and faunistic analysis of the site confirmed this point of view: all the remains at Dmanisi corresponded to a single sedimentation episode. These arguments were contested by those who recognized more than one species in the Dmanisi palaeodeme and who questioned the stratigraphy of the site but lacked the appropriate knowledge to correctly interpret it. However, for the authors of the study of the new skull, there was no doubt: it was an archaic population of our genus that, despite their limited cranial capacity, already showed characteristics derived of Homo erectus.
The reluctance to accept that the Dmanisi set corresponded to a single contemporary population is understandable because it entails revising much of the criteria used in Africa and other sites to interpret the human fossil record. The edge between Homo erectus and Homo ergaster, maintained for years, is diluted, as is the distinction between Homo habilis and Homo rudolfensis. The fact is that, unlike the multiple African findings, the Dmanisi palaeodeme corresponds to a single population. With five skulls and post-cranial remains of more than three individuals, it gives us an idea of the real variability of the first populations of the Homo genus (Lordkipanidze et al., 2007).
The emergence of palaeogenomics
From the end of the twentieth century to the present day, our interpretation of the fossil record has been shaken by the emergence of a new type of fossil, fossil DNA: a type of record that was unimaginable only a few decades ago. Sequencing mitochondrial DNA in organisms of the upper Pleistocene began successfully in the 1980s, and its application to Neanderthals, whose chronology fell within the conservation range of this genetic material, was a foregone conclusion. Interestingly, among the first Neanderthal remains to be analysed and which showed an acceptable level of conservation of the original DNA, were the fossils found in 1857 in the Neander valley, the ones that gave the name to the group. Subsequently, the sample of Neanderthals in which it was possible to purify authentic mitochondrial DNA was expanded to include the sites of La Chapelle-aux-Saints in France, Engis in Belgium, Mezmaiskaya in Russia, and Teshik-Tash in Uzbekistan. However, two sites – Vindija in Croatia and El Sidrón in Spain – stand out from the rest because of the level of conservation of their DNA, which is why they have contributed significantly to decoding this molecule in Neanderthals. Today, the recovery of mitochondrial DNA has been further extended to include the ancestors of Neanderthals, such as those from the Sima de los Huesos site in Atapuerca, whose human remains, attributed to Homo heidelbergensis, date back nearly 500,000 years.
Early results on mitochondrial DNA in Neanderthals revealed the existence of sequences that are very different from the current human type (Krings et al., 1997). In a total of 379 base pairs, 27 differences were observed, contrasting with the 5 or 8 differences expected for a random sample of current human DNA. This meant that the overall difference between Neanderthal sequences and modern humans was three times greater than the average among today’s humans, but only half of that observed between humans and chimpanzees. If we consider the divergence time between these latter two species, this implies that the divergence between the line that led to anatomically-modern man and the one that led to the Neanderthals is remarkably old: between 550,000 and 690,000 years ago. The fact that the lines leading to the two groups had evolved independently for more than half a million years was interpreted as clear evidence that Homo neanderthalensis and Homo sapiens constituted two different species.
This result put an end to the heated debates of the late twentieth century between supporters of the multiregional model, like Milford Wolpoff and Erik Trinkaus, and supporters of the «Noah’s Ark» model, such as Chris Stringer and Richard Klein, for explaining the origin of Homo sapiens. According to Wolpoff and Trinkaus, after the first departure from Africa, the different populations of Homo erectus would have continued to maintain a high genetic flow, so different regional populations would have developed the characteristics of Homo sapiens in parallel. Faced with this model, most palaeontologists advocated an exclusively African origin for our species, prior to its dispersion to the rest of the continents. The results provided by the analysis of the Neanderthal mitogenome confirmed this latter theory by revealing that Neanderthals and modern humans are different species. The next research step was the Neanderthal Genome Project, addressed jointly by two teams led by Richard E. Green, from the University of California, and Svante Pääbo, from the Max Plank Institute in Germany. This time the goal was no longer the reconstruction of mitochondrial DNA but rather to reconstruct the complete Neanderthals genome, as had previously been done with the human genome. After years of research, in which the Vindija and El Sidrón sites again played the most important role, a first draft was published in 2010 and included approximately 60% of the gene sequence (Green et al., 2010). The conclusions drawn in this publication were later confirmed in 2013 with the publication of the complete genome. Surprisingly, the analysis of Neanderthal DNA revealed the presence of 1-4% of Neanderthal sequences in the genome of modern Eurasian humans, but not in the genome of current African populations. This meant that this limited crossing must have taken place in the early stages of Homo sapiens’s expansion out of Africa, most likely when both populations coincided in the Palestine corridor between 40,000 and 60,000 years ago. Multiregionalists and proponents of cross-breeding between Neanderthals and modern humans welcomed this result because they interpreted it as an endorsement of their ideas. Strictly speaking, this implies that Neanderthals and sapiens cannot technically be considered different species.
For most paleoanthropologists, however, this result did not significantly alter the idea that Neanderthals and modern humans represent different species or quasi-species with a divergent evolutionary history spanning hundreds of thousands of years, especially considering that, after contact in Palestine, both populations had coexisted in Europe for more than 10,000 years without any new effective cross-breeding.
But the unveiling of the Neanderthal genome was not the only surprising result provided by palaeogenomics. Pääbo’s team carried out an analysis of a small fragment of a child’s phalanx from levels dating back 48,000 to 30,000 years from the Denisova Cave in the Altai Mountains of Siberia. It revealed a nucleotide sequence different not only from that of modern humans but also from that of Neanderthals (Krause et al., 2010). The Denisovans, as they were called, had inhabited Central Asia and had come across Neanderthals and modern humans. In fact, among modern populations, the Denisovan genome’s greatest concomitance was with the inhabitants of Papua New Guinea. However, no trace of Denisovan genes was found in much closer populations, such as the Chinese.
We are, therefore, faced with a hominin older than Neanderthals and modern humans, who coincided with them and even cross-bred. However, apart from a pair of large molars and the child’s phalanx, we know nothing about this mysterious hominin. The close relationship with Neanderthals would point to relict populations of Homo heidelbergensis, but they could correspond to even more archaic hominids, perhaps relict populations of Homo erectus, which at one point or another might have crossed paths with modern humans in Australasia. In any case, the emergence of Denisovans has ended up breaking the current moulds of paleoanthropological research.
REFERENCES
Agustí, J., & Lordkipanidze, D. (2005). Los primeros pobladores de Europa. Barcelona: RBA.
Green, R. E., Krause, J., Briggs, A. W., Maricic, T., Stenzel, U., Kircher, M., ... Pääbo, S. (2010). A draft sequence of the Neandertal genome. Science, 328, 710–722. doi: 10.1126/science.1188021
Krause, J., Fu, Q., Good, J. F., Viola, B., Shunkov, M. V., Derevianko, A. P., & Pääbo, S. (2010). The complete mitochondrial DNA genome of an unknown hominin from southern Siberia. Nature, 464, 894–897. doi: 10.1038/nature08976
Krings, M., Stone, A., Schmitz, R. W., Krainitzki, H., Stoneking, & Pääbo, S. (1997). Neandertal DNA sequences and the origin of modern humans. Cell, 90, 19–30. doi: 10.1016/S0092-8674(00)80310-4
Lordkipanidze, D., Vekua, A., Ferring, R., Rightmire, G. P., Agustí, J., Kiladze, G., … Zollikofer, C. (2005). The earliest toothless hominin skull. Nature, 434, 717–718. doi: 10.1038/434717b
Lordkipanidze, D., Jashashvili, T., Vekua, A., Ponce de León, M., Zollikofer, C., Rightmire, … Nioradze, M. (2007). Postcranial evidence from early Homo from Dmanisi, Georgia. Nature, 449, 305–310. doi: 10.1038/nature06134
Lordkipanidze, D., Ponce de León, M., Margvelashvili, A., Rak, Y., Rightmire, G. P., & Zollikofer, C. (2013). A complete skull from Dmanisi, Georgia, and the evolutionary biology of early Homo. Science, 342, 326–331. doi: 10.1126/science.1238484
Vekua, A., Lordkipanidze, D., Rightmire, G. P., Agustí, J., Ferring, R., Maisuradze, G., … Zollikofer, C. (2002). A new skull of early Homo from Dmanisi, Georgia. Science, 297, 85–89. doi: 10.1126/science.1072953
Villmoare, B., Kimbel, W. H., Seyoum, C., Campisano, C. J., DiMaggio, E., Rowan, J., … Reed, K. E. 2015. Early Homo at 2.8 Ma from Ledi Geraru, Afar, Ethiopia. Science, 347, 1352–1355. doi: 10.1126/science.aaa1343





